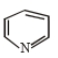
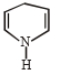
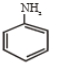
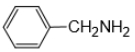
How many geometrical isomers are possible of the following?
CH3-CH=CH-CH=CH-CH3
1. 2 2. 3
3. 4 4. 6
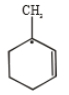
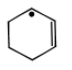
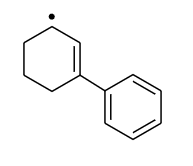
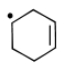
Most stable radical is
1. 
2. 
3. 
4. 
Which of the following is the most stable carbonium ion?
1.
2.
3.
4.
Which of the following will have minimum C-Cl bond length?
1. CH3-CH2-Cl
2. PhCH2-Cl
3. Ph-Cl
4. CH2=CH-CH2-Cl
The decreasing order of stability is-
1. A > B > C > D
2. D > B > C > A
3. D > C > B > A
4. A > B > D > C
The configuration of given tartaric acid is
1. 2R, 3R 2. 2R, 3S
3. 2S, 3S 4. 2S, 3R
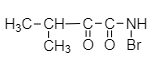
The IUPAC name of the above-mentioned compound is -
1. (N-Bromo)-3-methyl-2-oxobutanamide
2. (N-Bromo)-2-oxo-4-methylbutanamide
3. (N-Bromo)-1,2-dioxo-3-methylbutanamine carboxamide
4. (N-Bromo)-1-oxo-2-methylpropane
Which of the following -bonds participate in hyperconjugation?
| 1. | I and II | 2. | I and V |
| 3. | II and IV | 4. | III and IV |
Correct IUPAC name of the compound
1. 4-(Ethyl methanolyonxy)phenylpropanoate
2. Ethyl 4-propanoyloxybenzenecarboxylate
3. 4-(1-Oxo-2-oxabutyl)phenylpropanoate
4. 1-(1-Oxo-2-oxbutyl)-4-(1-oxopropoxy)benzene