Primary alcohols can be obtained from the reaction of the RMgX with:
1. HCHO
2. H2O
3. CO2
4. CH3CHO
CH3CH2ClX YZ
In the above reaction sequence, Z is
1. CH3CH2CH2NHCOCH3
2. CH3CH2CH2NH2
3. CH3CH2CH2CONHCH3
4. CH3CH2CH2CONHCOCH3
The best method to prepare cyclohexene from cyclohexanol is by using:
| 1. | conc. HCl + ZnCl2 | 2. | conc. H3PO4 |
| 3. | HBr | 4. | conc. HCl |
When 2-butanol is passed over heated copper at 573 K, it undergoes dehydrogenation to form:
1. 2-Butene
2. Butanone
3. Butyraldehyde
4. 1-Butene
Oxidation of 2-propanol by K2Cr2O7 and dilute H2SO4 leads to the formation of:
1. Propanal
2. Propanoic acid
3. Methanoic acid
4. Propanone
A compound X with molecular formula C3H8O can be oxidised to a compound Y with the molecular formula C3H6O2. X is most likely to be:
1. Primary alcohol
2. Secondary alcohol
3. Aldehyde
4. Ketone
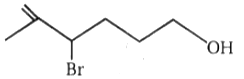
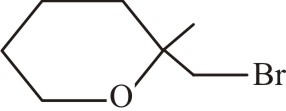
Consider the following alcohols,
| I. |  |
| II. |  |
| III. |  |
| IV. |  |
Ease of dehydration of the above alcohols will be in the order of:
| 1. | I < II < III < IV | 2. | I > II > III > IV |
| 3. | III < II < I < IV | 4. | II < III < IV < I |
R-CH2-CH2OH can be converted into RCH2CH2COOH. The correct sequence of reagents is:
1. PBr3, KCN, H+
2. PBr3, KCN, H2
3. KCN, H+
4. HCN, PBr3, H+
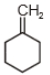
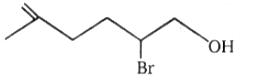
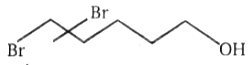
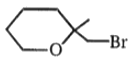
The major product in the acid catalysed dehydration of 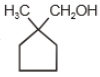 would be
would be
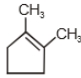
1. 
2. 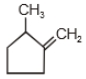
3. 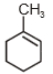
4. 
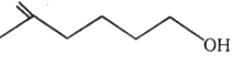

1. 
2. 
3. 
4.