The dihedral angle of the least stable conformer of ethane is:
1.
60°
2.
0°
3.
120°
4.
180°
The major product of the following chemical reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Reactant A in the following reaction is:
A + NaOH \(\xrightarrow[\Delta ]{CaO}\) CH3CH2CH3 + Na2CO3
1. Sodium ethanoate
2. Sodium propanoate
3. Sodium butanoate
4. Sodium methanoate
Which of the following is the correct balanced chemical equation for the combustion of butane?
| 1. | \(\mathrm{2 {C}_4 {H}_{10({g})}+13 {O}_{2({g})} \rightarrow 8 {CO}_{2({g})}+10 {H}_2 {O}} + Heat \) |
| 2. | \(\mathrm{2 {C}_4 {H}_{10({g})}+13 {O}_{2({g})} \rightarrow 9 {CO}_{2({g})}+10 {H}_2 {O}+} Heat \) |
| 3. | \(\mathrm{2 {C}_4 {H}_{10({g})}+14 {O}_{2({g})} \rightarrow 8 {CO}_{({g})}+10 {H}_2 {O}}+ Heat \) |
| 4. | \( \mathrm{{C}_4 {H}_{10({g})}+13 {O}_{2({g})} \rightarrow 8 {CO}_{({g})}+10 {H}_2 {O}}+ Heat\) |
The number of structural isomers of (with one double bond) and (with one triple bond) is:
1. \(\mathrm{C}_4 \mathrm{H}_8=3 ; \mathrm{C}_5 \mathrm{H}_8=3 \)
2. \(\mathrm{C}_4 \mathrm{H}_8=4 ; \mathrm{C}_5 \mathrm{H}_8=4 \)
3. \(\mathrm{C}_4 \mathrm{H}_8=3 ; \mathrm{C}_5 \mathrm{H}_8=4\)
4. \(\mathrm{C}_4 \mathrm{H}_8=4 ; \mathrm{C}_5 \mathrm{H}_8=3\)
The IUPAC names of the following compounds are?
(a) CH3CH=C(CH3)2
(b) CH2=CH-C≡C-CH3
(c) 
(d)
(e)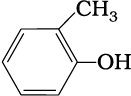
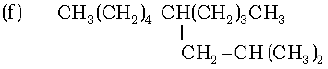
(g) CH3 – CH = CH – CH2 – CH = CH – CH – CH2 – CH = CH2
|
C2H5
1. 2-Methylbut-2-ene, Pen-1-ene-3-yne, 1, 3-Butadiene, 4-Phenyl but-1-ene , 2-Methyl phenol, 5-(2-Methylpropyl)-decane and 4-Ethyldeca-1, 5, 8-triene
2. 4-Phenyl but-1-ene, Pen-1-ene-3-yne, 1, 3-Butadiene, 2-Methylbut-2-ene, Pen-1-ene-3-yne, 2-Methyl phenol, 5-(2-Methylpropyl)-decane and 4-Ethyldeca-1, 5, 8-triene
3. 2-Methylbut-2-ene, 4-Phenyl but-1-ene, 1, 3-Butadiene, Pen-1-ene-3-yne, 2-Methyl phenol, 5-(2-Methylpropyl)-decane and 4-Ethyldeca-1, 5, 8-triene
4. 4-Ethyldeca-1, 5, 8-triene, Pen-1-ene-3-yne, 1, 3-Butadiene, 4-Phenyl but-1-ene, 2-Methyl phenol, 5-(2-Methylpropyl)-decane and 2-Methylbut-2-ene
Lewis acid(s) that can be used during the ethylation of benzene is/are:
| 1. |
|
2. |
|
| 3. |
|
4. | All of the above |
Which of the following will undergo nitration most easily: benzene, m-dinitrobenzene, or toluene?
| 1. | Benzene | 2. | m–Dinitrobenzene |
| 3. | Toluene | 4. | None of the above |
Benzene is extraordinarily stable, though it contains three double bonds, because of :
1. Delocalized protons
2. Delocalized neutrons
3. Resonance
4. None of the above.
Propanal and pentane-3-one are the products of ozonolysis of an alkene. The structural formula of the alkene is :
| 1. | 2. | ||
| 3. | 4. |  |







