The correct order of basicities of the following compounds is:
(1) 
(2)
(3)
(4)
1. 2>1>3>4
2. 1>3>2>4
3. 3>1>2>4
4. 1>2>3>4

The most reactive of these compounds towards sulphonation is
1. Toluene 2. Chlorobenzene
3. Nitrobenzene 4. m-Xylene
How many geometrical isomers are possible of the following?
CH3-CH=CH-CH=CH-CH3
1. 2 2. 3
3. 4 4. 6
The configuration of given tartaric acid is

1. 2R, 3R 2. 2R, 3S
3. 2S, 3S 4. 2S, 3R
Correct IUPAC name of the compound
1. 4-(Ethyl methanolyonxy)phenylpropanoate
2. Ethyl 4-propanoyloxybenzenecarboxylate
3. 4-(1-Oxo-2-oxabutyl)phenylpropanoate
4. 1-(1-Oxo-2-oxbutyl)-4-(1-oxopropoxy)benzene
The IUPAC name of the following compound is
1. 2-(Ethoxycarbonyl)benzoylchloride
2. Ethyl 2-(chlorocarbonyl)benzoate
3. Ethyl 2-(chloromethanoyl)benzoate
4. Methyl 2-(Chlorocarbonyl)benzene carboxylate.
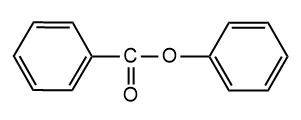 and
and 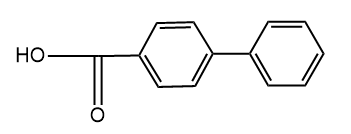 are
are
1. Position isomers
2. Chain isomers
3. Functional isomers
4. Metamers
Which of the following statements about the inductive effect is correct?
| 1. | The inductive effect transfers electrons from one carbon atom to another. |
| 2. | The inductive effect operates in both \(\sigma\)- and \(\pi\)-bonds. |
| 3. | The inductive effect does not create any charge in the molecule. |
| 4. | The inductive effect creates partial charges and is distance-dependent. |
The IUPAC name of the following compounds is:

1. 1-Bromocyclohexane carboxylic acid
2. 3-Bromocyclohexanoic acid
3. 3-Bromoheptanoic acid
4. 3-Bromocyclohexanecarboxylic acid
The correct increasing order of carbon-carbon bond length in the given compounds is:
1. III<II<I<IV
2. IV<I<II<III
3. I<II<III<IV
4. I<IV<III<II









