Pick out the incorrect statement
(1) Equivalent conductance increases with dilution
(2) Molar conductance increases with dilution
(3) Specific conductance increases with dilution
(4) Specific resistance increases with dilution
The cell potential for this reaction is 0.46V. Which of the following change will increase the potential the most ?
(1) concentration is doubled
(2) [Cu2+] concentration is halved
(3) Size of Cu(s) electrode is doubled
(4) size of Ag(s) electrode is decreased to half
What will be the value of for the rection
if
(1) -65.62 KJ
(2) -75.27 KJ
(3) -150.54 KJ
(4) +150.54 KJ
The cell reaction of micro electrochemical cell fromed during rusting of iron is:-
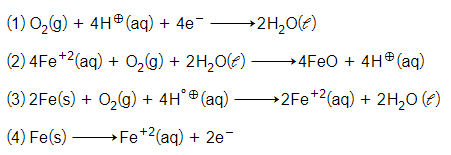
For the cell reaction
the change in free energy at a given temperature is a function of
(1) nC1
(2) n
(3) n (C1 +C2)
(4) nC2
The Emf of the following voltaic cell is:
\(\text {Ni }| \text {Ni}^{2+} (1 \text M) || \text {Au} ^{+3} \text {(1M) |Au}\)
\(\left ({~\mathrm{E}_{{\mathrm{Ni}^{+2}}/{\mathrm{Ni}}}^0=-0.25 \mathrm{~V}, \mathrm{E}_{{\mathrm{Au}^{+3}}/{\mathrm{Au}}}^0=1.50 \mathrm{~V}}\right )\)
1. 1.25 V
2. -1.75 V
3. 1.75 V
4. 4.0 V
The same amount of electric current is apassed through aqueous solution of MgSO4 and AlCl3. If 2.8 g Mg metal is deposited at amount of Al metal deposited in second cell will be
(1) 2.1 g
(2) 2.49 g
(3) 3.15 g
(4) 3.73g
What is the name of the device that converts the energy from the combustion of fuels such as hydrogen and methane directly into electrical energy?
(1) Fuel cell
(2) Electrolytic cell
(3) Dynamo
(4) Ni-Cd cell
gas can’t be obtained by the electrolysis of any salt because-
(A) Fluorine is the strongest reducing agent
(B) Fluorine is the strongest oxidising agent.
(C) Fluorine easily combine with atmospheric
(D) All
If the E°cell for a given reaction has a negative value then which of the following gives the correct relationships for the values of G° and Keq?
(a) G° < 0;Keq > 1
(b) G° < 0; Keq < 1
(c) G°> 0; Keq < 1
(d) G° > 0; Keq > 1




