

can be repared from williamson's synthesis using
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | All of these |
1. 
2. C6H5CH2Cl and
3. 
4. All of these
Methanol and ethanol can be distinguished by the following:
1. By reaction with metallic sodium
2. By reaction with caustic soda
3. By heating with iodine and washing soda
4. By heating with zinc and inorganic mineral acid
The product 'D' in the below mentioned reaction is:
\(CH_3MgBr + \text{cyclopentanone}\ \)
\(\xrightarrow{H_2O}\ A\ \xrightarrow{HBr}\ B\)
\( B\ \xrightarrow{Mg/Ether}\ C \xrightarrow[H_3O^+]{HCHO}\ D\)
| 1. |  |
2. | 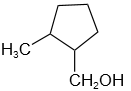 |
| 3. |  |
4. |  |
Chlorobenzene reacts with Mg in dry ether to give a compound (A) which further reacts
with ethanol to yield
1. phenol
2. benzene
3. ethyl benzene
4. phenyl ether
The compound B formed in the following sequence of reactions,
CH3CH2CH2OHAB will be:
1. Propyne
2. Propene
3. Propanal
4. Propane
The boiling point of ethanol is higher than that of dimethyl ether due to the presence of
| 1. | H-bonding in ethanol | 2. | H-bonding in dimethyl ether |
| 3. | -CH3 group in ethanol | 4. | -CH3 group in dimethyl ether |
The correct order of the acidic strength for the above compounds is:
 |
 |
 |
| (I) | (II) | (III) |
1. I > II > III
2. III > I > II
3. II > III > I
4. I > III > II
Tautomerism is not exhibited by-
1. 

2. 
3. 
4. 
Which of the following has highest boiling point?
1. Benzene
2. Phenol
3. Toluene
4. Ethyl benzene
p-nitrophenol is stronger acid than phenol because nitro group is:
1. Electron withdrawing
2. Electron donating
3. Basic
4. Acidic










