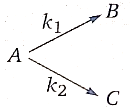
1. 757.48 K
2. 378.74 K
3. 600.91 K
4. None of the above
The activation energies of two reactions are and with > .
f the temperature of the reacting system is increased from T1 to T2 .
The correct relation is:
1.
2.
3.
4.
The units of rate constant and rate of reaction are same for :
1. First order reaction
2. Second order reaction
3. Third order reaction
4. Zero order reaction
The plot of log k vs helps to calculate :
1. Energy of activation.
2. Rate constant of reaction.
3. Order of the reaction.
4. Energy of activation as well as the frequency factor.
Which of the following represents the value of the pre-exponential factor (A) for a reaction when the rate constant is K = 1.2×103 mol–1 L s–1 and activation energy Ea = 2.0×102 kJ mol–1, in the limit \(T→∞\)?
1. 2.0 × 102 kJ mol–1
2. 1.2 × 103 mol–1L s–1
3. 1.2 × 103 mol L–1 s–1
4. 2.4 × 103 kJ mol–1L s–1
The rate constant for a reaction 2×10–2 s–1 at 300 K and 8×10–2 s–1 at 340 K. The energy of activation of the reaction is:
1. 14.69 kJ mol–1
2. 29.39 kJ mol–1
3. 44.34 kJ mol–1
4. 22.05 kJ mol–1
The reaction 2A + B + C D + E is found to be a first-order reaction with respect to A, second-order reaction with respect to B, and zero-order reaction with respect to C. If the concentrations of A, B, and C are doubled, the rate of the reaction will be:
1. 72 times
2. 8 times
3. 24 times
4. 36 times
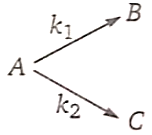
1. 100 kJ/mol
2. 120 kJ/mol
3. 116 kJ/mol
4. 220 kJ/mol


