The beans are cooked in pressure cooker, because -
1. Boiling point increases with increasing pressure
2. Boiling point decreases with increasing pressure
3. Extra pressure of pressure cooker softens the beans
4. Internal energy is not lost while cooking in pressure cooker
Boyle's law is applicable in :
1. Isobaric process
2. Isochoric process
3. Isothermal process
4. Adiabatic process
500 mL of nitrogen at 27C are cooled to -5C at the same pressure. The new volume becomes-
1. 326.32 mL
2. 446.66 mL
3. 546.32 mL
4. 771.56 mL
The number of molecules present in 1 mL of gas or vapor at STP is:
1. called Loschmidt's number
2. equal to 2.617 x 1019 per mL
3. both (1) and (2)
4. none of the above
Air at sea level is dense. This is a practical application of:
1. Boyle's law
2. Charles' law
3. Avogadro's law
4. Dalton's law
600 cc of a gas at a pressure of 750 mm is compressed to 500 cc. Taking the temperature to remain constant, the increase in pressure is:
1. 150 mm
2. 250 mm
3. 350 mm
4. 450 mm
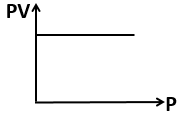
The curve that can not represent Boyle's law is -
1.
2.
3.
4.
The deviation of a gas from ideal behavior is expected to be minimum at -
1. 350 K and 3 atm
2. 550 K and 1 atm
3. 250 K and 4 atm
4. 450 K and 2 atm
An ideal gas expands according to PV=constant. On expansion, the temperature of the gas:
1. Will rise.
2. Will drop.
3. Will remain constant.
4. Cannot be determined because the external pressure is not known.
The correct statement for an ideal gas equation is -
1. n is the number of molecules of a gas.
2. V denotes the volume of 1 mole of the gas.
3. n moles of a gas has a volume V.
4. p is the pressure of the gas when only one mole of the gas is present.