Which position will be attacked most rapidly by the nitronium ion (-NO2)+ when the compound undergoes nitration with HNO3/H2SO4 :-
1. A
2. B
3. C
4. D
Which of the following substitution of benzene is ortho-para in electrophilic substitution and ortho-para in nucleophilic substitution ?
(1) -NO2
(2) -NO
(3) -SO3H
(4) -SO2Me
Which one of the following undergoes nucleophilic aromatic substitution at the fastest rate?
(1)
(2)
(3)
(4)
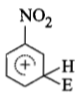
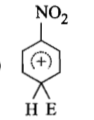
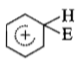
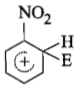
The electrophile. E attacks the benzene ring to generate the intermediate - complex. Of the following which - complex is of lowest energy?
1. 
2. 
3. 
4. 
Which of the following meta-directing substituents is the most deactivating in aromatic substitution?
| 1. | –SO3H | 2. | –COOH |
| 3. | –NO2 | 4. | –C≡N |
Among cyclohexanol (I), acetic acid (II), 2, 4, 6-trinitrophenol (III) and phenol (IV) the correct order of decreasing acidic character will be:
| 1. | III>II>IV>I | 2. | II>III>I>IV |
| 3. | II>III>IV>I | 4. | III>IV>II>I |
Following is the list of some aromatic compounds. Select the correct sequence of decreasing order of reactivity for electrophilic aromatic substitution reaction using the answer codes given below:
a.
b.
c.
d.
Answer codes :
1. a d b c
2. c d b a
3. a b d c
4. a b c d
Consider the following statements :
a. group is o, p-directing group due to hyperconjugation.
b. is meta directing group due to reverse hyperconjugation.
c. group is meta directing group due to mesomeric effect.
d. CHO group is m-directing group
Of these statements :
1. a and b are correct
2. a and c are correct
3. a, b and d are correct
4. c and d are correct
Arrange the reactivity of given compounds in decreasing order for hydrolysis reaction?
a.
b.
c.
d.
Select the correct answer form the codes given below:
Codes :
1. a b c d
2. b c d a
3. b c a d
4. a d b c
Which alcohol is most reactive for dehydrogenation or dehydration?
1.
2.
3.
4.











