Transition state structure of the substrate formed during an enzymatic reaction is
1. Transient but stable
2. Permanent but unstable
3. Transient but unstable
4. Permanent and stable
The substrate has to go through a much higher energy state or transition state in case of:
| 1. | Exothermic reactions only |
| 2. | Endothermic reactions only |
| 3. | Both exothermic and endothermic reactions |
| 4. | Neither exothermic nor endothermic reactions |
Which of the following describes the graph correctly?
1. Endothermic reaction with energy A in the presence of enzyme and B in the absence of enzyme
2. Exothermic reaction with energy A in the presence of enzyme and B in the absence of enzyme
3. Endothermic reaction with energy A in the absence of enzyme and B in the presence of enzyme
4. Exothermic reaction with energy A in the absence of enzyme and B in the presence of enzyme
Activation energy:
1. Is the difference in the average energy content of 'S' from that of its transition state
2. Helps to change reactants into unstable transition state before they can be converted into products
3. Is minimum energy required from outside to overcome the energy barrier of reactant or to start a reaction
4. All are correct
I. Segment representing the energy of activation.
II. Segment representing the amount of free energy released by the reaction.
III. Transition state
IV. Segment would be the same regardless of whether the reaction were uncatalysed or catalysed
Which one is correct?
|
|
I |
II |
III |
IV |
|
(a) |
1 |
3 |
2 |
4 |
|
(b) |
1 |
2 |
3 |
2 |
|
(c) |
1 |
3 |
2 |
4 |
|
(d) |
1 |
2 |
4 |
3 |
The figure given below shows the conversion of a substrate into product by an enzyme. In which one of the four options (a-d) the components of reaction labelled as A, B, C and D are identified correctly?
|
A |
B |
C |
D |
|
|
(1) |
Potential energy |
Transition state |
Activation energy with enzyme |
Activation energy without enzyme |
|
(2) |
Transition state |
Potential energy |
Activation energy without enzyme |
Activation energy with enzyme |
|
(3) |
Potential energy |
Transition state |
Activation energy with enzyme |
Activation energy without enzyme |
|
(4) |
Activation energy with enzyme |
Transition state |
Activation energy without enzyme |
Potential energy |
Which of the following is correct?
1. Physical change refers to change in shape without breaking the bonds
2. Change in state of matter also takes place in physical change
3. Chemical change involves breaking of old bonds and formation of new ones.
4. All of these
Which of the following is incorrect?
| 1. | The ES complex formation is a transient phenomenon |
| 2. | The structure of the substrate gets transformed into the structure of product. |
| 3. | All the structures formed between substrate and product are called intermediates. |
| 4. | Intermediates are highly stable. |
Which of the following is not true for the graph?
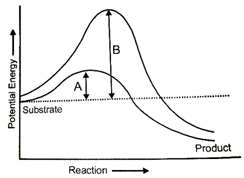
1. The reaction is exothermic
2. The reaction is endothermic
3. The product is more stable than substrate
4. The difference in average energy content of substrate from that of transition state is called activation energy.
Which of the following is correct for enzyme catalysed reaction?
1. Substrate is conveted into Enzyme
2. Substrate is consumed in the reaction completely
3. Substrate is converted to product
4. Enzyme is converted to product









