A compound A of formula C3H6Cl2 on reaction with alkali can give B of formula C3H6O or C of formula C3H4. B on oxidation gave a compound of the formula C3H7O2. C with dilute H2SO4 containing Hg2+ ion gave D of formula C3H6O, which with bromine and NaOH gave the sodium salt of C2H4O2. Then A is :
1. CH3CH2CHCl2
2. CH3CCl2CH3
3. CH2ClCH2CH2Cl
4. CH3CHClCH2Cl
Identify the product (A) in following reaction series, (X)(Y)(Z)(A) :
1. CH3CHO
2. CH3CONH2
3. CH3COOH
4. CH3 - CH2-NHOH
Compound (A), C8H9Br , gives a white precipitate when warmed with alcholic AgNO3. Oxidation of (A) gives an acid (B), C8H6O4. (B) easily forms anhydride on heating. Identify the compound (A).
| 1. |   |
| 2. | 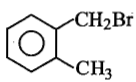  |
| 3. |   |
| 4. |   |
Which of the following will give yellow precipitate with I2/NaOH?
| 1. | CH3-CO-O-CO-CH3 | 2. |  |
| 3. |  |
4. | Both (2) and (3) |
A nitrogeneous substance X is treated with HNO2 and the product so formed is further treated with NaOH solution, which produces blue colouration. X can be
1. CH3CH2NH2
2 CH3CH2NO2
3. CH3CH2ONO
4 (CH3)2CHNO2
The compound which reacts fastest with Lucas reagent at room temperature is:
1. Butan-1-ol
2. Butan-2-ol
3. 2-methylpropan-1-ol
4. 2-methylpropan-2-ol
Identify (X) in the sequence :
1. CH3-CH2-CH2OH
2.
3. CH3-O-CH2-CH3
4. CH3-CH2-CHO
In order to accomplish the following conversion, what reagent and conditions would be required?
1. Cold sodium hydroxide
2. Hot conc. sodium hydroxide
3. Potassium tertiary butoxide and heat
4. Hot water
An amine is reacted with benzene sulphonyl chloride then a solid compound is formed which is insoluble in alkali. The amine is:
1. CH3-CH2-NH2
2. CH3-NH-CH2-CH3
3. (CH3)3N
4. C6H5-NH2
Nitrobenzene on electrolytic reduction in a strongly acidic medium gives:
1. Aniline
2. p-Aminophenol
3. m-Nitroaniline
4. Nitrosobenzene







