What is the correct order of the methyl-substituted amines' basic strength in an aqueous solution?
1. CH3NH2 > (CH3)2NH > (CH3)3N
2. (CH3)2NH > CH3NH2 > (CH3)3N
3. (CH3)3N > CH3NH2 > (CH3)2NH
4. (CH3)2NH > (CH3)3N > CH3NH2
Which of the following compounds has a higher basic character than aniline?
1. Diphenylamine
2. Triphenylamine
3. p-nitroaniline
4. Benzylamine
Propanoic acid gives a series of reactions as given below.
The structure of D would be:
1.
2.
3.
4.
Which compound, upon reduction with lithium aluminium hydride, yields a secondary amine?
1. Nitroethane
2. Methylisocyanide
3. Acetamide
4. Methyl cyanide
The structure of C in the below-mentioned reaction is:
| 1. | 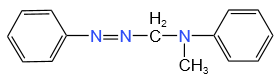 |
| 2. |  |
| 3. |  |
| 4. |  |
What is the product formed in the below reaction?

| 1. |  |
2. |  |
| 3. |  |
4. |  |
The incorrect statement among the following regarding the primary amine is:
| 1. | Alkyl amines are stronger bases than aryl amines |
| 2. | Alkyl amines react with nitrous acid to produce alcohols |
| 3. | Aryl amines react with nitrous acid to produce phenols |
| 4. | Alkyl amines are stronger bases than ammonia |
The reagent that is used to convert acetamide into methyl amine is:
1. NaOH-Br2
2. Soda lime
3. Hot conc. H2SO4
4. PCl5
Aniline gives a set of the following reactions that yielded a coloured product 'Y':

The structure of 'Y' is :
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Nitrobenzene on reaction with conc. HNO3 /H2SO4 at 80 – 100 ºC forms:
1. 1,3– Dinitrobenzene
2. 1,4–Dinitrobenzene
3. 1,2,4–Trinitrobenzene
4. 1,2–Dinitrobenzene







