Select Chapter Topics:
What is the correct order of the methyl-substituted amines' basic strength in an aqueous solution?
1. CH3NH2 > (CH3)2NH > (CH3)3N
2. (CH3)2NH > CH3NH2 > (CH3)3N
3. (CH3)3N > CH3NH2 > (CH3)2NH
4. (CH3)2NH > (CH3)3N > CH3NH2
Subtopic: Amines - Preparation & Properties |
77%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Which amine reacts with Hinsberg's reagent to produce an alkali insoluble product?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
78%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Which amine gives the carbylamine test?
| 1. | 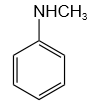 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
86%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The reaction of propanamide with ethanolic sodium hydroxide and bromine will give:
| 1. | Ethylamine | 2. | Methylamine |
| 3. | Propylamine | 4. | Aniline |
Subtopic: Amines - Preparation & Properties |
74%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Identify the compound that will react with Hinsberg's reagent to give a solid which dissolves in alkali.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Identification of Primary, Secondary & Tertiary Amines |
80%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The reagent 'R' in the given sequence of a chemical reaction is :

| 1. | HI | 2. | CuCN/KCN |
| 3. | H2O | 4. | CH3CH2OH |
Subtopic: Diazonium Salts: Preparation, Properties & Uses |
57%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| Statement I: | Primary aliphatic amines react with HNO2 to give unstable diazonium salts. |
| Statement II | Primary aromatic amines react with HNO2 to form diazonium salts which are stable even above 300 K. |
| 1. | Statement I is incorrect but Statement II is correct |
| 2. | Both Statement I and II are correct |
| 3. | Both Statement I and II are incorrect |
| 4. | Statement I is correct but Statement II is incorrect |
Subtopic: Diazonium Salts: Preparation, Properties & Uses |
52%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The product formed from the following reaction sequence is


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Diazonium Salts: Preparation, Properties & Uses | Mechanism |
75%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Match list-I with list-II:
| List-I (Amines) | List-II (pKb values) | ||
| (a) | N-Methylmethanamine | (i) | 9.30 |
| (b) | Ammonia | (ii) | 9.38 |
| (c) | N-Methylaniline | (iii) | 4.75 |
| (d) | Benzenamine | (iv) | 3.27 |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (ii) | (i) | (iii) |
| 2. | (iv) | (iii) | (i) | (ii) |
| 3. | (iii) | (iv) | (i) | (ii) |
| 4. | (i) | (iv) | (iii) | (ii) |
Subtopic: Amines - Preparation & Properties |
70%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The major product (P) formed in the following reaction sequence is :
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Amines - Preparation & Properties |
69%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital







