Select Question Set:
Identify the correct reagents that would bring about the following transformation.


| 1. | \(\text { (i) } \mathrm{BH}_3\) | 2. | \(\text { (i) } \mathrm{BH}_3\) |
| \((ii) \mathrm{H}_2 \mathrm{O}_2 / \stackrel{\ominus}{\mathrm{OH}}\) | \((ii) \mathrm{H}_2 \mathrm{O}_2 / \stackrel{\ominus}{\mathrm{OH}}\) | ||
| \(\text { (iii) } \mathrm{P C C}\) | \(\text { (iii) alk. } \mathrm{KMnO}_4\) | ||
| \(\text { (iv) } \mathrm{H}_3 \mathrm{O}^{+}\) | |||
| 3. | \(\text { (i) } \mathrm{H}_2 \mathrm{O} / \mathrm{H}^{+}\) | 4. | \(\text { (i) } \mathrm{H}_2 \mathrm{O} / \mathrm{H}^{+}\) |
| \(\text { (ii) } \mathrm{P C C}\) | \(\text { (ii) } \mathrm{CrO}_3\) |
Subtopic: Alcohols: Preparation & Properties |
64%
Level 2: 60%+
NEET - 2024
Hints
Which one of the following alcohols reacts instantaneously with Lucas reagent?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | \(\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{OH}\) |
Subtopic: Alcohols: Preparation & Properties |
82%
Level 1: 80%+
NEET - 2024
Hints
Consider the following reaction and identify the product (P):


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Alcohols: Preparation & Properties |
73%
Level 2: 60%+
NEET - 2023
Hints
Which amongst the following will be most readily dehydrated under acidic conditions?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Mechanism of Dehydration, Methanol & Ethanol |
54%
Level 3: 35%-60%
NEET - 2023
Hints
Consider the following reaction:

Identify products A and B:

Identify products A and B:
| 1. |  |
| 2. | 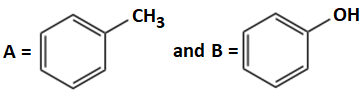 |
| 3. |  |
| 4. |  |
Subtopic: Ethers: Preparation & Properties, Uses |
74%
Level 2: 60%+
NEET - 2023
Hints
Reagents that can be used to convert alcohols to carboxylic acids are:
(A) CrO3 - H2SO4
(B) K2Cr2O7 + H2SO4
(C) KMnO4 + KOH/H3O+
(D) Cu, 573 K
(E) CrO3+ (CH3CO)2O
Choose the most appropriate answer from the options given below:
1. (B), (C) and (D) only
2. (B), (D) and (E) only
3. (A), (B) and (C) only
4. (A), (B) and (E) only
(A) CrO3 - H2SO4
(B) K2Cr2O7 + H2SO4
(C) KMnO4 + KOH/H3O+
(D) Cu, 573 K
(E) CrO3+ (CH3CO)2O
Choose the most appropriate answer from the options given below:
1. (B), (C) and (D) only
2. (B), (D) and (E) only
3. (A), (B) and (C) only
4. (A), (B) and (E) only
Subtopic: Alcohols: Preparation & Properties |
72%
Level 2: 60%+
NEET - 2023
Hints
Consider the following reactions:
\({R-COOH\xrightarrow[(ii)~H_2O/HCl]{(i)~X}R-CH_2OH }\)
\(\small{R-CH=CH_2\xrightarrow[(ii)~H_2O/ NaOH/ H_2O_2]{(i)~X}R-CH_2-CH_2-OH}\)
Identify 'X' in above reactions:
\({R-COOH\xrightarrow[(ii)~H_2O/HCl]{(i)~X}R-CH_2OH }\)
\(\small{R-CH=CH_2\xrightarrow[(ii)~H_2O/ NaOH/ H_2O_2]{(i)~X}R-CH_2-CH_2-OH}\)
Identify 'X' in above reactions:
| 1. | \(B_2H_6 \) | 2. | \(LiAlH_4 \) |
| 3. | \(NaBH_4 \) | 4. | \(H_2/Pd \) |
Subtopic: Alcohols: Preparation & Properties |
51%
Level 3: 35%-60%
NEET - 2023
Hints
The major product formed in the following conversion is:


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Alcohols: Preparation & Properties | Mechanism of Dehydration, Methanol & Ethanol |
65%
Level 2: 60%+
NEET - 2023
Hints
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| I: | The acidic strength of monosubstituted nitrophenol is higher than phenol because of electron withdrawing nitro group. |
| II: | o-Nitrophenol, m-nitrophenol and p-nitrophenol will have same acidic strength as they have one nitro group attached to the phenolic ring. |
| 1. | I is incorrect but II is correct. |
| 2. | Both I and II are correct. |
| 3. | Both I and II are incorrect. |
| 4. | I is correct but II is incorrect. |
Subtopic: Phenols: Preparation & Properties |
84%
Level 1: 80%+
NEET - 2022
Hints
Given below are two statements:
In light of the above statements, choose the most appropriate answer from the options given below:
| Statement I: | In Lucas test, primary, secondary, and tertiary alcohols are distinguished on the basis of their reactivity with conc. \(HCl + ZnCl_2\), known as Lucas Reagent. |
| Statement II: | Primary alcohols are most reactive and immediately produce turbidity at room temperature on reaction with Lucas Reagent. |
| 1. | I is incorrect but II is correct |
| 2. | Both I and II correct |
| 3. | Both I and II are incorrect |
| 4. | I is correct but II is incorrect |
Subtopic: Alcohols: Preparation & Properties |
81%
Level 1: 80%+
NEET - 2022
Hints
Select Question Set:






