An organic compound A reacts with sodium metal and forms B. On heating with conc. H2SO4, A gives diethyl ether. So A and B are:
1. C3H7OH and CH3ONa
2. CH3OH and CH3ONa
3. C4H9OH and C4H9ONa
4. C2H5OH and C2H5ONa
| 1. | Lead compounds in liquor | 2. | Methyl alcohol in liquor |
| 3. | Ethyl alcohol in liquor | 4. | Carbonic acid in liquor |
Upon dehydration, the below mentioned compound will not give:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Consider the following alcohols,
| I. |  |
| II. |  |
| III. |  |
| IV. |  |
Ease of dehydration of the above alcohols will be in the order of:
| 1. | I < II < III < IV | 2. | I > II > III > IV |
| 3. | III < II < I < IV | 4. | II < III < IV < I |
The best method to prepare cyclohexene from cyclohexanol is by using:
| 1. | conc. HCl + ZnCl2 | 2. | conc. H3PO4 |
| 3. | HBr | 4. | conc. HCl |

| A | B | |
| 1. |  |
 |
| 2. | 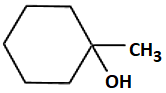 |
 |
| 3. |  |
 |
| 4. |  |
 |
What is the correct order of dehydration rate for the compounds (i), (ii), and (iii) when treated with concentrated \(\text{H}_2\text{SO}_4\)?
| (i) |  |
| (ii) |  |
| (iii) |  |
1. (i) > (iii) > (ii)
2. (i) > (ii) > (iii)
3. (ii) > (i) > (iii)
4. (ii) > (iii) > (i)
Optically active 2-octanol rapidly loses its optical activity when exposed to :
| 1. | Dilute acid | 2. | Dilute base |
| 3. | Light | 4. | Humidity |
The most reactive compound among the following toward electrophilic aromatic substitution reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The proposed mechanism for hydration of ethene to yield ethanol is as follows:

The wrong step in the above mechanism is:
| 1. | Step 1 | 2. | Step 2 |
| 3. | Step 3 | 4. | Both steps 2 and 3 |





