Shown below are adsorption isotherms for a gas 'X' at temperatures T1 , T2 and T3 :
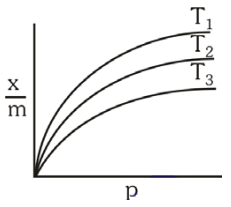
p and \(\frac xm\) represent pressure and extent of adsorption, respectively. The correct order of temperatures for the given adsorption is:
1. T1 > T2 > T3
2. T3 > T2 > T1
3. T1 = T2 = T3
4. T1 = T2 > T3

p and \(\frac xm\) represent pressure and extent of adsorption, respectively. The correct order of temperatures for the given adsorption is:
1. T1 > T2 > T3
2. T3 > T2 > T1
3. T1 = T2 = T3
4. T1 = T2 > T3
Subtopic: Adsorption and Absorption |
68%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The characteristics associated with adsorption are:
1. ∆G, ∆H, and ∆S are all negative.
2. ∆G and ∆H are negative, but ∆S is positive.
3. ∆G and ∆S are negative, but ∆H is positive.
4. ∆G is negative, but ∆H and ∆S are positive.
Subtopic: Adsorption and Absorption |
81%
From NCERT
NEET - 2016
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Which of the following statements is correct for the spontaneous adsorption of a gas?
| 1. | ∆ S is negative and therefore, ∆ H should be highly positive |
| 2. | ∆ S is negative and therefore, ∆ H should be highly negative |
| 3. | ∆ S is positive and therefore, ∆ H should be negative |
| 4. | -∆ S is positive and therefore, ∆ H should also be highly positive |
Subtopic: Adsorption and Absorption |
55%
From NCERT
AIPMT - 2014
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
A plot of log x/m versus log p for the adsorption of a gas on a solid gives a straight line with a slope equal to:
1. log
2. n
3.
4. log 1/k
Subtopic: Adsorption and Absorption | Adsorption Isotherm |
78%
From NCERT
AIPMT - 2006
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
According to the adsorption theory of catalysis, the speed of the reaction increase because:
| 1. | The concentration of reactant molecules at the active centers of the catalyst becomes high due to adsorption |
| 2. | In the process of adsorption, the activation energy of the molecules becomes large |
| 3. | Adsorption produces heat which increases the speed of the reaction |
| 4. | Adsorption lowers the activation energy of the reaction |
Subtopic: Adsorption and Absorption |
From NCERT
AIPMT - 2003
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Which statement is not correct regarding the adsorption of a gas on the surface of a solid:
| 1. | On increasing temperature adsorption increase continuously |
| 2. | Enthalpy & entropy change is negative |
| 3. | Adsorption is more for some specific substance |
| 4. | Reversible |
Subtopic: Adsorption and Absorption |
75%
From NCERT
AIPMT - 2001
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital




