Select Chapter Topics:
Which one is an example of heterogeneous catalysis?
| 1. | Combination between dinitrogen and dihydrogen to form ammonia in the presence of finely divided iron. |
| 2. | Oxidation of sulphur dioxide into sulphur trioxide in the presence of oxides of nitrogen. |
| 3. | Hydrolysis of sugar catalysed by H+ ions. |
| 4. | Decomposition of ozone in presence of nitrogen monoxide. |
Subtopic: Catalyst |
54%
From NCERT
NEET - 2023
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Pumice stone is an example of-
| 1. | foam | 2. | sol |
| 3. | gel | 4. | solid sol |
Subtopic: Colloidal Solution |
61%
From NCERT
NEET - 2023
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Which of the following is a positively charged sol?
1. Methylene blue sol
2. Congo red sol
3. Siver sol
4. \(Sb_2S_3 ~sol\)
1. Methylene blue sol
2. Congo red sol
3. Siver sol
4. \(Sb_2S_3 ~sol\)
Subtopic: Colloidal Solution |
50%
From NCERT
NEET - 2023
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The incorrect statement regarding enzymes is:
| 1. | Enzymes are very specific for a particle reaction and substrate. |
| 2. | Enzymes are biocatalysts. |
| 3. | Like chemical catalysts, enzymes reduce the activation energy of bio processes. |
| 4. | Enzymes are polysaccharides. |
Subtopic: Catalyst |
79%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| Statement I: | In the coagulation of a negative sol, the flocculating power of the three given ions is in the order Al3+ > Ba2+ > Na+ |
| Statement II: | In the coagulation of a positive sol, the flocculating power of the three given salts is in the order NaCl > Na2SO4 > Na3PO4 |
| 1. | I is incorrect but II is correct. |
| 2. | Both I and II are correct. |
| 3. | Both I and II are incorrect. |
| 4. | I is correct but II is incorrect. |
Subtopic: Colloidal Solution |
66%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Match List-I with List-II:
| List-I (Example of Colloidal systems) |
List-II (Nature of dispersion medium and dispersed phase) |
| (a) Insecticide spray | (i) Dispersion medium - liquid Dispersed phase - solid |
| (b) Whipped cream | (ii) Dispersion medium - gas Dispersed phase - liquid |
| (c) Paint | (iii) Dispersion medium - liquid Dispersed phase - liquid |
| (d) Hair cream | (iv) Dispersion medium - liquid Dispersed phase -gas |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (iv) | (ii) | (iii) | (i) |
| 3. | (ii) | (iv) | (iii) | (i) |
| 4. | (iv) | (ii) | (i) | (iii) |
Subtopic: Colloidal Solution |
52%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Shown below are adsorption isotherms for a gas 'X' at temperatures T1 , T2 and T3 :
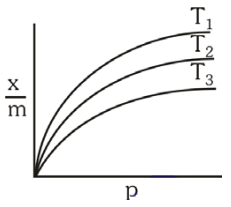
p and \(\frac xm\) represent pressure and extent of adsorption, respectively. The correct order of temperatures for the given adsorption is:
1. T1 > T2 > T3
2. T3 > T2 > T1
3. T1 = T2 = T3
4. T1 = T2 > T3

p and \(\frac xm\) represent pressure and extent of adsorption, respectively. The correct order of temperatures for the given adsorption is:
1. T1 > T2 > T3
2. T3 > T2 > T1
3. T1 = T2 = T3
4. T1 = T2 > T3
Subtopic: Adsorption and Absorption |
68%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The right option for the statement "Tyndall effect is exhibited by", is:
1. Starch solution
2. Urea solution
3. NaCl solution
4. Glucose solution
Subtopic: Colloidal Solution |
65%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Measuring Zeta potential is useful in determining which of the following property of a colloidal solution:
1. Solubility.
2. Stability of the colloidal particles.
3. Size of the colloidal particles.
4. Viscosity.
Subtopic: Colloidal Solution |
77%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
In which of the sols, the colloidal particles are with a negative charge?
1. TiO2
2. Hemoglobin
3. Starch
4. Hydrated Al2O3
Subtopic: Colloidal Solution |
53%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital




