The graph between vs log p is given below:
What is the value of the slope?
1.
2. n
3.
4.
Subtopic: Adsorption Isotherm |
91%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
In the Freundlich adsorption isotherm, the value of 1/n is:
1. Between 0 and 1 in all cases.
2. Between 2 and 4 in all cases.
3. 1 in the case of physical adsorption.
4. 1 in the case of chemisorption.
Subtopic: Adsorption Isotherm |
91%
From NCERT
AIPMT - 2012
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
An adsorption isotherm is a plot between:
| 1. | The extent of adsorption \(({x \over m})\) against the pressure of gas (P) at constant V. |
| 2. | The extent of adsorption \(({x \over m})\) against the pressure of gas (P) at constant T. |
| 3. | The extent of absorption \(({x \over m})\) against the volume of gas (V) at constant P. |
| 4. | The extent of absorption \(({x \over m})\) against the pressure of gas (P) at constant T. |
Subtopic: Adsorption Isotherm |
81%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The equation that represents a Freundlich adsorption isotherm is:
1.
2.
3.
4.
Subtopic: Adsorption Isotherm |
71%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
The correct curve for Freundlich's adsorption isotherm is:
| 1. | 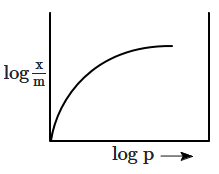 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Adsorption Isotherm |
64%
From NCERT
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital
Hints
Links
To view explanation, please take trial in the course.
NEET 2026 - Target Batch - Vital



