If NaCl is doped with 10-4 mol % of SrCl2, the concentration of cation vacancies will be
(NA = 6.023 x 1023 mol-1)
1. 6.023 x 1015 mol-1
2. 6.023 x 1016 mol-1
3. 6.023 x 1017 mol-1
4. 6.023 x 1014 mol-1
The phenomenon in which polar crystals on heating produce electricity is called:-
1. Pyro-electricity.
2. Piezo-electricity.
3. Ferro-electricity.
4. Ferri-electricity.
Total volume of atoms present in a face centred cubic unit cell of a metal is -
(r is atomic radius)
1. 20/3 r3
2. 24/3 r3
3. 12/3 r3
4. 16/3 r3
Lithium borohydride (LiBH4), crystallizes in an orthorhombic system with 4 molecules per unit cell. The unit cell dimensions are:- a = 6.81 Å , b = 4.43 Å, c = 7.17 Å. If the molar mass of LiBH4 is 21.76 g mol-1, then the density of the crystal is:-
1. 0.67 g cm-3
2. 0.58 g cm-3
3. 1.23 g cm-3
4. None of the above.
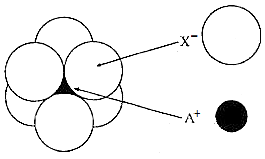
The arrangement of X- ions around A+ ion in solid AX is given in the figure (not drawn to scale).
If the radius of X- is 250 pm, the radius of A+ is:
1. 104 pm
2. 125 pm
3. 183 pm
4. 57 pm
In an f.c.c. unit cell. atoms are numbered as shown below. The atoms not touching each other are (Atom numbered 3 is face centre of front face).
1. 3&4
2. 1&3
3. 1&2
4. 2&4
A solid has a bcc structure. If the distance of closest approach between the two atoms is 1.73 Å, then the edge length of the cell is:
1. 200 pm
2. \(\frac{\sqrt{3}}{\sqrt{2}}\) pm
3. 142.2 pm
4. \(\sqrt{2 }\)pm
Which is covalent solid?
1. Silicate
2. Diamond
3. Graphite
4. All of these
What type of crystal defects is indicated in the diagram given below?
1. Frenkel and Schottky defects
2. Schottky defect
3. Interstitial defect
4. Frenkel defect
The anions (A) form hexagonal closest packing and atoms (C) occupy only 2/3 of octahedral voids
in it. The general formula of the compound is-
1. CA
2. CA2
3. C2A3
4. C3A2