Predict the correct intermediate and product in the following reaction.
H3C-CCH Intermediate Product
(A) (B)


The correct order of acidity among the following is:
| 1. | \(\mathrm{CH \equiv CH > CH_3 - C \equiv CH }\) \(\mathrm{ > CH_2 = CH_2 > CH_3 - CH_3}\) |
| 2. | \(\mathrm{CH \equiv CH > CH_2 = CH_2 }\) \(\mathrm{ > CH_3 - C \equiv CH > CH_3 - CH_3}\) |
| 3. | \(\mathrm{CH_3 - CH_3 > CH_2 = CH_2 }\) \(\mathrm{ > CH_3 - C \equiv CH > CH \equiv CH }\) |
| 4. | \(\mathrm{ CH_2 = CH_2 > CH_3 - CH_3 }\) \(\mathrm{ > CH_3 - C \equiv CH > CH \equiv CH}\) |

1. \(A :\) \(H_{3} C - \underset{OH}{\underset{\left|\right.}{C}} = CH_{2}\) \(\) \(;\) \(B :\) \(\) \(H_{3} C - \underset{HSO_{4}}{\underset{\left|\right.}{C}} = CH_{2}\)
2. \(A :\) \(H_{3} C - \underset{O}{\underset{\left|\right. \left|\right.}{C}} - CH_{3}\) \(\) \(;\) \(B :\) \(\) \(H_{3} C - C \equiv CH\)
3. \(A :\) \(H_{3} C - \overset{OH}{\overset{\left|\right.}{C}} = CH_{2}\) \(\) \(;\) \(B :\) \(\) \(H_{3} C - \overset{O}{\overset{\left|\right. \left|\right.}{C}} - CH_{3}\)
4. \(A :\) \(H_{3} C - \underset{HS O_{4}}{\underset{\left|\right.}{C}} = CH_{2}\) \(\) \(;\) \(B :\) \(\) \(H_{3} C - \underset{O}{\underset{\left|\right. \left|\right.}{C}} - CH_{3}\)
Which of the following compounds shall not produce propene by reaction with HBr followed by elimination or direct only elimination reaction?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
In the given reaction

The product P is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The compound that will react most readily with gaseous bromine has the formula:
| 1. | \(\mathrm{C_3H_6}\) | 2. | \(\mathrm{C_2H_2}\) |
| 3. | \(\mathrm{C_4H_{10}}\) | 4. | \(\mathrm{C_2H_4}\) |
Hydrocarbon (A) reacts with bromine by substitution reaction to form an alkyl bromide B.
B undergoes the Wurtz reaction to give a gaseous hydrocarbon containing less than four carbon atoms.
The formula of (A) is:
1.
2.
3.
4.
The compound undergoes the following reactions
C
The product 'C' is-
1. m–Bromotoluene
2. o–Bromotoluene
3. 3–Bromo–2,4,6–trichlorotoluene
4. p–Bromotoluene
P, Q and R in the above-mentioned sequence of reactions are respectively:
| 1. |  |
| 2. |  |
| 3. | 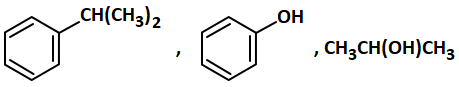 |
| 4. |  |
The molecule among the following that has the hybridization from left to right atoms is:
1.
2.
3.
4.







