If 300 ml of a gas at 27°C is cooled to 7°C at constant pressure, its final volume will be:
| 1. | 135 ml | 2. | 540 ml |
| 3. | 350 ml | 4. | 280 ml |
A plot of volume versus temperature (T) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in the figure given below.
The correct order of pressure is -
Temperature (K)
1.
2.
3.
4.
At 0°C, the density of a certain oxide of a gas at 2 bar is the same as that of dinitrogen at 5 bar.
The molecular mass of the oxide would be -
1. 35 g/mol
2. 45 g/mol
3. 70 g/mol
4. 60 g/mol
The plots of different values of pressure versus temperature are shown in the given figure. The correct order of volume for the following plot is-
1. V4 < V3 < V2 < V1
2. V4 > V3 < V2 > V1
3. V4 > V3 > V2 > V1
4. V4 > V3 > V2 < V1
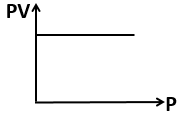
The variation of pressure with the volume of the gas at different temperatures can be
graphically represented as per the following graph:
If the temperature increases from 200K to 400K (at constant pressure), the volume of a
gas would-
1. Increase
2. Decrease
3. Remain constant
4. 1st decreases then increases
The density of methane at 2.0 atmosphere pressure and 27°C is-
1. 0.13 gL–1
2. 0.26 gL–1
3. 1.30 gL–1
4. 2.60 gL–1
The deviation of a gas from ideal behavior is expected to be minimum at -
1. 350 K and 3 atm
2. 550 K and 1 atm
3. 250 K and 4 atm
4. 450 K and 2 atm