The element Z = 114 has been discovered recently. Family/group and electronic configuration from which it will belong are:
1. Carbon family and [Rn] 5f14 6d10 7s2 7p2
2. Oxygen family and [Rn] 5f14 6d10 7s2 7p4
3. Nitrogen family and [Rn] 5f14 6d10 7s2 7p6
4. Halogen family and [Rn] 5f14 6d10 7s2 7p5
1. Carbon family and [Rn] 5f14 6d10 7s2 7p2
2. Oxygen family and [Rn] 5f14 6d10 7s2 7p4
3. Nitrogen family and [Rn] 5f14 6d10 7s2 7p6
4. Halogen family and [Rn] 5f14 6d10 7s2 7p5
How many electrons can fit in the subshell for which n = 3 and l = 1?
| 1. | 2 | 2. | 6 |
| 3. | 10 | 4. | 14 |
Incorrect statement among the following is:
| 1. | The uncertainty principle is\(\Delta x \cdot \Delta p \geq \dfrac{h}{4 \pi} \) |
| 2. | Half-filled and fully filled orbitals have greater stability due to greater exchange energy, greater symmetry, and a more balanced arrangement. |
| 3. | The energy of the 2s orbital is less than the energy of the 2p orbital in the case of hydrogen-like atoms. |
| 4. | De-Broglie's wavelength is given by\(\lambda=\dfrac{h}{m v} \), where m= mass of the particle, v = group velocity of the particle. |
A pair of d-orbitals having electron density along the axes is:
1.
2.
3.
4.
The incorrect statement among the following is:
| 1. | Total orbital angular momentum of an electron in 's' orbital is equal to zero. |
| 2. | An orbital is designated by three quantum numbers, while an electron in an atom is designated by four quantum numbers. |
| 3. | The electronic configuration of N atom is 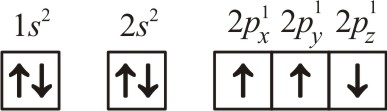 |
| 4. | The value of m for dz2 is zero. |
Two electrons occupying the same orbital are distinguished by:
1. Magnetic quantum number
2. Azimuthal quantum number
3. Spin quantum number
4. Principal quantum number
Which is the correct order of increasing energy of the listed orbitals in the atom of titanium?
1. 3s 4s 3p 3d
2. 4s 3s 3p 3d
3. 3s 3p 3d 4s
4. 3s 3p 4s 3d
What is the maximum number of orbitals that can be identified with the following quantum numbers?
n = 3, l = 1, m = 0
| 1. | 1 | 2. | 2 |
| 3. | 3 | 4. | 4 |
Calculate the energy corresponding to light of wavelength 45 nm:
(Planck's constant h = 6.63 × 10–34 Js:
speed of light c = 3 × 108 ms–1)
1. 6.67 x 1015 J
2. 6.67 x 1011 J
3. 4.42 x 10-15 J
4. 4.42 x 10-18 J
1. 25
2. 50
3. 75
4. 10




