The circumference of the Bohr orbit for the H atom is related to the de Broglie wavelength associated with the electron revolving around the orbit by the following relation:
1.
2.
3.
4.
The largest de Broglie wavelength among the following (all have equal velocity) is:
1. molecule
2. molecule
3. Electron
4. Proton
The wavelength of an electron moving with a velocity of 2.05 × 107 m s–1 would be:
\(1 .\) \(4 . 65\) \(\times\) \(10^{- 12}\) \(m\)
\(2 .\) \(3 . 55\) \(\times\) \(10^{-11}\) \(m\)
\(3 .\) \(2 . 34\) \(\times\) \(10^{11}\) \(m\)
\(4 .\) \(6 . 43\) \(\times\) \(10^{ -11}\) \(m\)
If the velocity of the electron is 1.6 × 106 . The de Broglie wavelength associated with this electron is:
Identify the graph that correctly depicts the variation of momentum with the de-Broglie wavelength of a particle.
| 1. |  |
2. | 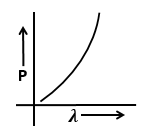 |
| 3. | 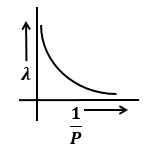 |
4. | 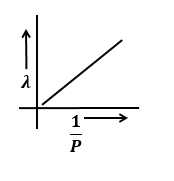 |
Incorrect statement among the following is:
| 1. | The uncertainty principle is\(\Delta x \cdot \Delta p \geq \dfrac{h}{4 \pi} \) |
| 2. | Half-filled and fully filled orbitals have greater stability due to greater exchange energy, greater symmetry, and a more balanced arrangement. |
| 3. | The energy of the 2s orbital is less than the energy of the 2p orbital in the case of hydrogen-like atoms. |
| 4. | De-Broglie's wavelength is given by\(\lambda=\dfrac{h}{m v} \), where m= mass of the particle, v = group velocity of the particle. |
The velocity associated with a proton moving at a potential difference of 1000 V is 4.37 × 105 ms–1. If the hockey ball of mass 0.1 kg is moving with this velocity, then the wavelength associated with this velocity would be:
The kinetic energy of an electron is \(3.0 \times 10^{-25}~ \mathrm J.\) Its wave length would be:
1. \(8.96 \times 10^{-7}~ \mathrm m\)
2. \(4.37 \times 10^{-6}~ \mathrm m\)
3. \(1.32 \times 10^{-7}~ \mathrm m\)
4. \(2.89 \times 10^{-4}~ \mathrm m\)
What is the velocity associated with a neutron that has a wavelength of 800 pm?
| 1. | 496 m s–1 | 2. | 567 m s–1 |
| 3. | 494 cm s–1 | 4. | 501 m s–1 |






