The orbital diagram in which both Pauli's exclusion principle and Hund's rule are violated is:
1.
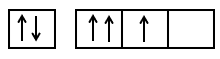
2.

3.

4.





The correct order of the total number of nodes of atomic orbitals is:
1. 4f > 6s > 5d
2. 6s > 5d > 4f
3. 4f > 5d > 6s
4. 5d > 4f > 6s
Correct statement among the following is:
1. Number of angular nodes = n - l - 1
2. Number of radial nodes = l
3. Total number of nodes = n -1
4. All of the above
Orbitals having two spherical nodes are:
| 1. | 2s | 2. | 4s |
| 3. | 3d | 4. | 6f |
The development of the wave mechanical model of the atom is based on which of the following concepts?
1. De-Broglie concept of the dual nature of the electron.
2. Heisenberg uncertainty principle.
3. Schrodinger's principle
4. All of the above
The incorrect statement regarding the characteristics of X-rays is :
1. The radiation can ionize the gas
2. It causes a fluorescence effect on ZnS
3. It is deflected by electric and magnetic fields
4. Have a shorter wavelength than ultraviolet rays
For any H-like system, the ratio of velocities of I, II & III orbit i.e., will be:
1. 1 : 2 : 3
2. 1 : 1/2 : 1/3
3. 3 : 2 : 1
4. 1 : 1 : 1
In photoelectric effect, the kinetic energy of photoelectrons increases linearly with the:
1. Wavelength of incident light.
2. Frequency of incident light.
3. Velocity of incident light.
4. Atomic mass of an element.
The photoelectric emission from a surface starts only when the light incident upon the surface has a certain minimum:
| 1. | Intensity | 2. | Wavelength |
| 3. | Frequency | 4. | Velocity |
The ratio of slopes of curves in photoelectric effect gives -
(v=frequency, =maximum kinetic energy, stopping potential):
1. Charge of electron
2. Planck's constant
3. Work function
4. The ratio of Planck's constant of electronic charge






