The number of unpaired electrons in 1s2 2s2 2p3 is -
1. 2
2. 0
3. 3
4. 1
ion and a proton are accelerated by the same potential. The ratio of their de-Broglie wavelengths is
(assume mass of proton = mass of neutron)
1. 1:2
2. 1:4
3. 1:1
4. 1:
| Assertion (A): | All d orbitals are tetra-lobed structures having two nodal planes. |
| Reason (R): | Out of five, only four d-orbitals are degenerate orbitals. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
The incorrect statement about the nodal plane among the following is:
| 1. | A plane on which there is a zero probability of finding an electron. |
| 2. | A plane on which there is maximum probability that the electron will be found. |
| 3. | ψ2 is zero at nodal plane. |
| 4. | None of the above. |
The quantum number not obtained from Schrodinger’s wave equation is:
| 1. | \(n\) | 2. | \(l\) |
| 3. | \(m\) | 4. | \(s\) |
A dye absorbs a photon of wavelength and re-emits the same energy into two photons of wavelengths and respectively. The wavelength is related to and as:
1.
2.
3.
4.
The excited state of an H atom emits a photon of wavelength and returns to the ground state. The principal quantum number of the excited state is given by:
1.
2.
3.
4.
Identify the graph that correctly depicts the variation of momentum with the de-Broglie wavelength of a particle.
| 1. |  |
2. | 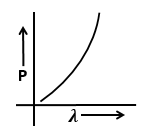 |
| 3. | 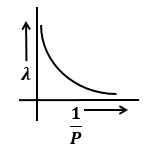 |
4. | 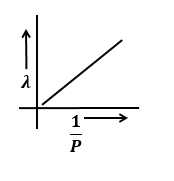 |
The largest de Broglie wavelength among the following (all have equal velocity) is:
1. molecule
2. molecule
3. Electron
4. Proton
The ratio of slopes of curves in photoelectric effect gives -
(v=frequency, =maximum kinetic energy, stopping potential):
1. Charge of electron
2. Planck's constant
3. Work function
4. The ratio of Planck's constant of electronic charge






