I. n=4; l=2; \(m_l\)=-2; s=-\(\frac1{2}\)
II. n=3; l=2; \(m_l\)=1; s=+\(\frac1{2}\)
III. n=4; l=1; \(m_l\)=0; s=+\(\frac1{2}\)
IV. n=3; l=1; \(m_l\)=-1; s=+\(\frac1{2}\)
The correct decreasing order of energy of these electrons is:
1. IV>II>III>I
2. I>III>II>IV
3. III>I>II>IV
4. I>II>III>IV
Sorry!! currently, the explanation for the question is not provided. If you need further help, please email at support@neetprep.com with subject: Explanation Missing for Question Id: 468860
Sorry!! currently, the explanation for the question is not provided. If you need further help, please email at support@neetprep.com with subject: Explanation Missing for Question Id: 468860
Which one of the following electrons in the ground state will have least amount of energy?
| 1. | An electron in hydrogen atom. |
| 2. | An electron in 2p orbital of carbon atom. |
| 3. | The electron of copper atom present in 4s orbital. |
| 4. | The outermost electron in sodium atom. |
4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy. The correct option is:
| 1. | 5f > 6p > 4d > 5p | 2. | 5f > 6p > 5p > 4d |
| 3. | 6p > 5f > 5p > 4d | 4. | 6p > 5f > 4d > 5p |
The incorrect statement among the following is:
| 1. | Total orbital angular momentum of an electron in 's' orbital is equal to zero. |
| 2. | An orbital is designated by three quantum numbers, while an electron in an atom is designated by four quantum numbers. |
| 3. | The electronic configuration of N atom is 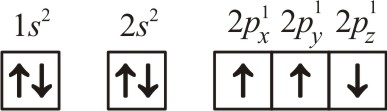 |
| 4. | The value of m for dz2 is zero. |
The element Z = 114 has been discovered recently. Family/group and electronic configuration from which it will belong are:
1. Carbon family and [Rn] 5f14 6d10 7s2 7p2
2. Oxygen family and [Rn] 5f14 6d10 7s2 7p4
3. Nitrogen family and [Rn] 5f14 6d10 7s2 7p6
4. Halogen family and [Rn] 5f14 6d10 7s2 7p5
How many electrons can fit in the subshell for which n = 3 and l = 1?
| 1. | 2 | 2. | 6 |
| 3. | 10 | 4. | 14 |
Which is the correct order of increasing energy of the listed orbitals in the atom of titanium?
1. 3s 4s 3p 3d
2. 4s 3s 3p 3d
3. 3s 3p 3d 4s
4. 3s 3p 4s 3d
If n = 6, the correct sequence for the filling of electrons will be:
1.
2.
3.
4.




