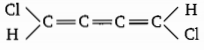The compound that has the lowest boiling point is:
| 1. | HF | 2. | HCl |
| 3. | HBr | 4. | HI |
The correct statement about hydrolysis of and is-
1. is hydrolysed and gives but is not hydrolysed
2. Both and on hydrolysis gives
3. on hydrolysis gives but gives
4. Both and on hydrolysis gives
The correct sequence of the polarity of the following molecule is
a. Benzene
b. Inorganic Benzene
c.
d.
| a | b | c | d | |
| 1. | P | NP | NP | P |
| 2. | NP | NP | NP | P |
| 3. | NP | P | NP | P |
| 4. | NP | P | P | NP |
(Where P=polar, NP=non-polar)
The incorrect order of boiling point is-
1.
2.
3.
4.
Which of the following is not a perfect nonpolar molecule?
1.
2. CCl4
3.
4. All of the above
Choose the correct option for the following molecule:
1. Non planar
2.
3. Both (1) and (2)
4.
The compound that has the highest ionic character associated with the X-Cl bond is:
1.
2.
3.
4.
The least polar bond is present in which of the following?
| 1. | O-F | 2. | P-F |
| 3. | Si-N | 4. | B-F |
The correct order of increasing s-character (in percentage) in the hybrid orbitals of the following molecules/ions is:
| (i) | \(\mathrm{CO}^{2-}_3\) |
| (ii) | \(\mathrm{XeF}_4\) |
| (iii) | \(\mathrm{I}^-_3\) |
| (iv) | \(\mathrm{NCl}_3\) |
| (v) | \(\mathrm{BeCl}_2\) |
| 1. | (ii)<(iii)<(iv)<(i)<(v) | 2. | (ii)<(iv)<(iii)<(v)<(i) |
| 3. | (iii)<(ii)<(i)<(v)<(iv) | 4. | (ii)<(iv)<(iii)<(i)<(v) |