In the following pairs of halogen compounds, the pair that would undergo SN2 reaction faster-
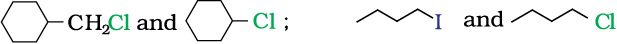
1.


2.


3.


4.


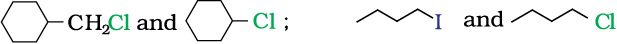








Correct statement among the following is-
| a. | For SN2 reactivity order is CH3CH2CH2CH2Br > (CH3)2CHCH2Br > CH3CH2CH(Br)CH3 > (CH3)3CBr |
| b. | For SN1 reactivity order is (CH3)3CBr > CH3CH2CH(Br)CH3 >CH3CH2CH2CH2Br >(CH3)2CHCH2Br |
| c. | For SN1 reactivity order is C6H5CH2Br < C6H5CH(CH3)Br < C6H5CH(C6H5)Br< C6H5C(CH3)(C6H5)Br |
| d. | For SN2 reactivity order is C6H5CH2Br > C6H5CH(CH3)Br > C6H5CH(C6H5)Br > C6H5C(CH3)(C6H5)Br |
The correct statement(s) is/are-
1. a, b
2. b, c
3. a, c, d
4. b, d
The incorrect matches among the following are:
| a. |  |
| b. |  |
| c. |  |
| d. |  |
| 1. | a, b | 2. | b, c, d |
| 3. | c, d | 4. | c, b |
| Assertion (A): | chlorine is an electron-withdrawing group, yet it is ortho-, para- directing in electrophilic aromatic substitution reactions. |
| Reason (R): | Halogen atom is a ring deactivator. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
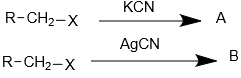
Consider the following reaction.
The product A and B are respectively-
1. A= R-CH2-CN; B= R-CH2-NC
2. A= R-CH2-NC; B= R-CH2-CN
3. A= R-NCH-CH3; B= R-CH2-CN
4. A= R-CH2-NC; B= R-CHN-CH3
Consider the following reaction
The product A is-
| 1. |  |
2. |  |
| 3. |  |
4. | None of the above |
The number of monochloro structural isomers formed on free radical monochlorination of (CH3)2CHCH2CH3 is-
1. 5
2. 3
3. 4
4. 6
Match the items in column I with the column II.
| Column I: Structures | Column II: IUPAC name |
a.  |
i. 4-Bromo- 3-methylpent-2-ene |
b.  |
ii. 3-Bromo-2-methylpropene |
c.  |
iii. 1-Bromo-2-methylbut-2-ene |
d.  |
iv. 4-Bromopent-2-ene |
1. a=iii; b=iv; c=i; d=ii
2. a=ii; b=iv; c=i; d=iii
3. a=iv; b=i; c=ii; d=iii
4. a=iv; b=ii, c=iv; d=iii
The correct statement among the following about C5H11Br is-
| 1. | It has total 8 isomers, 4 are primary alkyl bromide isomers and 2 are tertiary alkyl bromide isomers. |
| 2. | It has total 7 isomers, 4 are primary alkyl bromide isomers and 2 are tertiary alkyl bromide isomers. |
| 3. | It has total 8 isomers, 4 are primary alkyl bromide isomers and 3 are secondary alkyl bromide isomers. |
| 4. | It has total 7 isomers, 3 are secondary alkyl bromide isomers and 1 is a tertiary alkyl bromide isomer. |
The product A is-
1. 2-Chloropropane
2. 2-Chlorobutane
3. 1-Chlorobutane
4. 3-chloropropane