1. Probability of collision.
2. Orientation of reactant molecules during collision.
3. Rate constant at two different temperatures.
4. Rate constant at standard temperature.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Given R = 8.314 J K-1 mol-1, log 4 = 0.6021
1. 380.4 kJ/mol
2. 3.80 kJ/mol
3. 3804 kJ/mol
4. 38.04 kJ/mol
A: energy greater than threshold energy.
B: breaking of old bond in reactant.
C: formation of new bond in product.
D: high activation energy.
E: proper orientation.
Choose the correct answer from the options given below:
1. A, B, C, D only
2. A, B, C, E only
3. A, C, D, E only
4. B, C, D, E only
| Assertion (A): | A reaction can have zero activation energy. |
| Reason (R): | The minimum extra amount of energy absorbed by reactant molecules so that their energy becomes equal to threshold value, is called activation energy. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
The plot of ln k vs \({1 \over T}\) for the following reaction
\(2N_2O_5(g) \rightarrow 4NO_2 (g) + O_2(g) \) gives a straight line with the slope of the line equal to \(-1.0 \times 10^4 K \).
The activation energy for the reaction in J mol–1 is:
(Given R = 8.3 J K–1 mol–1)
1. \(4.0 \times 10^2 \)
2. \(4.0 \times 10^{-2} \)
3. \(8.3 \times 10^{-4} \)
4. \(8.3 \times 10^4 \)
For a reaction AB, enthalpy of reaction is and enthalpy of activation is . The correct potential energy profile for the reaction is:
| 1. | 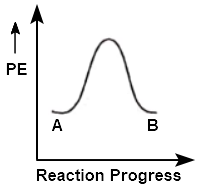 |
2. |  |
| 3. |  |
4. |  |
The slope of Arrhenius Plot (ln k v/s ) of the first-order reaction is . The value of Ea of the reaction is:
[Given R = 8.314 JK–1 mol–1]
| 1. | 166 kJ mol–1 | 2. | –83 kJ mol–1 |
| 3. | 41.5 kJ mol–1 | 4. | 83.0 kJ mol–1 |
An increase in the concentration of the reactants of a reaction leads to a change in:
1. heat of reaction
2. threshold energy
3. collision frequency
4. activation energy
What does ZAB represent in the collision theory of chemical reactions?
1. The fraction of molecules with energies greater than Ea
2. The collision frequency of reactants, A and B
3. Steric factor
4. The fraction of molecules with energies equal to Ea


