Which of the following graph shows the variation of pressure P with volume V for an ideal gas at a constant temperature?
1.

2.

3.

4.
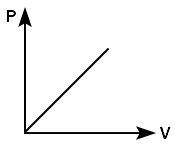




A gas performs the minimum work when it expands:
| 1. | Isochorically | 2. | Isobarically |
| 3. | Adiabatically | 4. | Isothermally |
An ideal gas goes from \(A\) to \(B\) via two processes, \(\mathrm{I}\) and \(\mathrm{II},\) as shown. If \(\Delta U_1\) and \(\Delta U_2\) are the changes in internal energies in processes \(\mathrm{I}\) and \(\mathrm{II},\) respectively, (\(P:\) pressure, \(V:\) volume) then:

| 1. | \(∆U_1 > ∆U_2\) | 2. | \(∆U_1 < ∆U_2\) |
| 3. | \(∆U_1 = ∆U_2\) | 4. | \(∆U_1 \leq ∆U_2\) |
An ideal monoatomic gas \(\left(\gamma = \frac{5}{3}\right )\) absorbs 50 cal in an isochoric process. The increase in internal energy of the gas is:
| 1. | 20 cal | 2. | Zero |
| 3. | 50 cal | 4. | 30 cal |
The pressure-temperature \((P\text-T)\) graph for two processes, \(A\) and \(B,\) in a system is shown in the figure. If \(W_1\) and \(W_2\) are work done by the gas in process \(A\) and \(B\) respectively, then:

| 1. | \(W_{1}=W_2\) | 2. | \(W_{1}<W_2\) |
| 3. | \(W_{1}>W_2\) | 4. | \(W_{1}= - W_2\) |
The variation of molar heat capacity at constant volume with temperature T for a monatomic gas is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
When a system is moved from state \(a\) to state \(b\) along the path \(acb\), it is discovered that the system absorbs \(200~\text{J}\) of heat and performs \(80~\text{J}\) of work. Along the path \(adb\), heat absorbed \(Q =144~\text{J}\). The work done along the path \(adb\) is:

| 1. | \(6~\text{J}\) | 2. | \(12~\text{J}\) |
| 3. | \(18~\text{J}\) | 4. | \(24~\text{J}\) |
1. \(5:3\)
2. \(5:2\)
3. \(1:1\)
4. \(5:7\)
In the cyclic process shown in the pressure-volume \((P-V)\) diagram, the change in internal energy is equal to:
1.
2.
3.
4. zero
1. \(20~\text{J}\)
2. \(0~\text{J}\)
3. \(-20~\text{J}\)
4. \(-15~\text{J}\)





