1 kg of gas does 20 kJ of work and receives 16 kJ of heat when it is expanded between two states. The second kind of expansion can be found between the same initial and final states, which requires a heat input of 9 kJ. The work done by the gas in the second expansion will be:
1.
32 kJ
2.
5 kJ
3.
-4 kJ
4.
13 kJ
The figure below shows two paths that may be taken by a gas to go from state A to state C. In process AB, \(400~\text{J}\) of heat is added to the system and in process BC, \(100~\text{J}\) of heat is added to the system. The heat absorbed by the system in the process AC will be:
| 1. | \(380~\text{J}\) | 2. | \(500~\text{J}\) |
| 3. | \(460~\text{J}\) | 4. | \(300~\text{J}\) |
The latent heat of vaporisation of water is \(2240~\text{J/gm}\). If the work done in the process of expansion of \(1~\text{g}\) is \(168~\text{J}\),
then the increase in internal energy is:
1. \(2408~\text{J}\)
2. \(2240~\text{J}\)
3. \(2072~\text{J}\)
4. \(1904~\text{J}\)
1. \(\frac{\eta}{2}\)
2. \(\eta\)
3. \(2\eta\)
4. \(3\eta\)
A monoatomic ideal gas, initially at temperature \(T_1\), is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature \(T_2\) by releasing the piston suddenly. If \(L_1\) and \(L_2\) are the lengths of the gas column before and after expansion, respectively, then \(\frac{T_1}{T_2}\) is given by:
1. \(\left(\frac{L_1}{L_2}\right)^{\frac{2}{3}}\)
2. \(\frac{L_1}{L_2}\)
3. \(\frac{L_2}{L_1}\)
4. \(\left(\frac{L_2}{L_1}\right)^{\frac{2}{3}}\)
The initial pressure and volume of a gas are \(P\) and \(V\), respectively. First, it is expanded isothermally to volume \(4V\) and then compressed adiabatically to volume \(V\). The final pressure of the gas will be: [Given: \(\gamma = 1.5\)]
| 1. | \(P\) | 2. | \(2P\) |
| 3. | \(4P\) | 4. | \(8P\) |
If \(n\) moles of an ideal gas is heated at a constant pressure from \(50^\circ\text C\) to \(100^\circ\text C,\) the increase in the internal energy of the gas will be:
\(\left(\frac{C_{p}}{C_{v}} = \gamma\ ~\text{and}~\ R = \text{gas constant}\right)\)
| 1. | \(\dfrac{50nR}{\gamma - 1}\) | 2. | \(\dfrac{100nR}{\gamma - 1}\) |
| 3. | \(\dfrac{50n\gamma R}{\gamma - 1}\) | 4. | \(\dfrac{25n\gamma R}{\gamma - 1}\) |
The first law of thermodynamics is based on:
| 1. | the concept of temperature. |
| 2. | the concept of conservation of energy. |
| 3. | the concept of working of heat engine. |
| 4. | the concept of entropy. |
The efficiency of an ideal heat engine is less than \(100\%\) because of:
| 1. | the presence of friction. |
| 2. | the leakage of heat energy. |
| 3. | unavailability of the sink at zero kelvin. |
| 4. | all of these. |
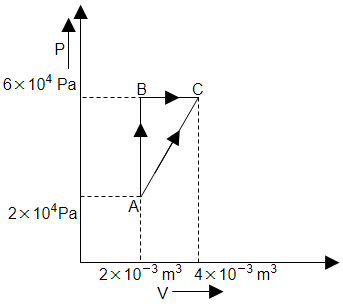
Work done during the given cycle is:
1. 4
2. 2
3.
4.





