
| 1. | \(4\) | 2. | \(1\) |
| 3. | \(2\) | 4. | \(3\) |
| 1. | \(\text{If}~P_1>P_2~\text{then}~T_1<T_2\) |
| 2. | \(\text{If}~V_2>V_1~\text{then}~T_2>T_1\) |
| 3. | \(\text{If}~V_2>V_1~\text{then}~T_2<T_1\) |
| 4. | \(\text{If}~P_1>P_2~\text{then}~V_1>V_2\) |
Two cylinders \(A\) and \(B\) of equal capacity are connected to each other via a stop cock. A contains an ideal gas at standard temperature and pressure. \(B\) is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is:
1. adiabatic
2. isochoric
3. isobaric
4. isothermal
The \((P\text{-}V)\) diagram for an ideal gas in a piston-cylinder assembly undergoing a thermodynamic process is shown in the figure. The process is:
| 1. | adiabatic | 2. | isochoric |
| 3. | isobaric | 4. | isothermal |
| 1. | isochoric | 2. | isothermal |
| 3. | adiabatic | 4. | isobaric |
Thermodynamic processes are indicated in the following diagram:
Match the following:
| Column-I | Column-II | ||
| (P) | Process I | (a) | Adiabatic |
| (Q) | Process II | (b) | Isobaric |
| (R) | Process III | (c) | Isochoric |
| (S) | Process IV | (d) | Isothermal |
| 1. | P → c, Q → a, R → d, S→ b |
| 2. | P→ c, Q → d, R → b, S → a |
| 3. | P → d, Q → b, R → b, S → c |
| 4. | P → a, Q → c, R → d, S → b |
A gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then,
| 1. | compressing the gas through an adiabatic process will require more work to be done. |
| 2. | compressing the gas isothermally or adiabatically will require the same amount of work. |
| 3. | which of the case (whether compression through isothermal or through the adiabatic process) requires more work will depend upon the atomicity of the gas. |
| 4. | compressing the gas isothermally will require more work to be done. |
| 1. | \(64P\) | 2. | \(32P\) |
| 3. | \(\frac{P}{64}\) | 4. | \(16P\) |
1. \(2\)
2. \(5/3\)
3. \(3/2\)
4. \(4/3\)
One mole of an ideal gas goes from an initial state \(A\) to the final state \(B\) with two processes. It first undergoes isothermal expansion from volume \(V\) to \(3V\) and then its volume is reduced from \(3V\) to \(V\) at constant pressure. The correct \((P-V)\) diagram representing the two processes is:
| 1. |  |
2. | 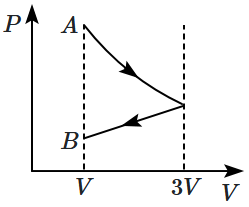 |
| 3. |  |
4. |  |








