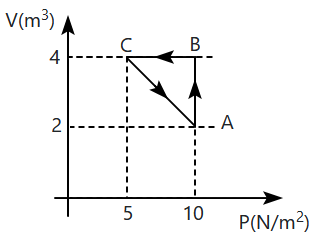
If in the thermodynamic process shown in the figure, the work done by the system along A B C is 50 J and the change in internal energy during C A is 30 J, then the heat supplied during A B C is:

1.
50 J
2.
20 J
3.
10 J
4.
80 J

The pressure in a monoatomic gas increases linearly from 4 atm to 8 atm when its volume increases from 0.2 m to 0.5 m. The increase in internal energy will be:
| 1. | 480 kJ | 2. | 550 kJ |
| 3. | 200 kJ | 4. | 100 kJ |
The degree of freedom per molecule for a gas on average is 8. If the gas performs 100 J of work when it expands under constant pressure, then the amount of heat absorbed by the gas is:
1. 500 J
2. 600 J
3. 20 J
4. 400 J
An ideal gas is taken through the cycle \(A\rightarrow B\rightarrow C\rightarrow A\) as shown in the figure below. If the net heat supplied to the gas is \(10~\text{J}\), then the work done by the gas in the process \(B\rightarrow C\) is:

| 1. | \(-10~\text{J}\) | 2. | \(-30~\text{J}\) |
| 3. | \(-15~\text{J}\) | 4. | \(-20~\text{J}\) |
An ideal gas undergoes a cyclic process ABCA as shown. The heat exchange between the system and the surrounding during the process will be:
| 1. | 10 J | 2. | 5 J |
| 3. | 15 J | 4. | 20 J |
If the ratio of specific heat of a gas at constant pressure to that at constant volume is , the change in internal energy of a mass of gas, when the volume changes from V to 2V at constant pressure, P is:
| 1. | 2. | PV | |
| 3. | 4. |
An ideal gas is taken through the process as shown in the figure. Then:
| 1. | In the process AB, the work done by the system is positive. |
| 2. | In process AB, heat is rejected out of the system. |
| 3. | In the process AB, internal energy increases. |
| 4. | In the process AB, internal energy decreases, and in the process BC, internal energy increases. |
\(ABCA\) is a cyclic process. Its \(P\text-V\) graph would be:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
In the \((P\text-V)\) diagram shown, the gas does \(5~\text J\) of work in the isothermal process \(ab\) and \(4~\text J\) in the adiabatic process \(bc.\) What will be the change in internal energy of the gas in the straight path from \(c\) to \(a?\)

1. \(9~\text J\)
2. \(1~\text J\)
3. \(4~\text J\)
4. \(5~\text J\)
A horizontal cylinder has two sections of unequal cross-sections in which two pistons, A and B, can move freely. The pistons are joined by a string. Some gas is trapped between the pistons. If this gas is heated, the pistons will:
| 1. | move to the left. |
| 2. | move to the right. |
| 3. | remain stationary. |
| 4. | move either to the left or to the right depending on the initial pressure of the gas. |