| 1. | \(64P\) | 2. | \(32P\) |
| 3. | \(\frac{P}{64}\) | 4. | \(16P\) |
1. \(2\)
2. \(5/3\)
3. \(3/2\)
4. \(4/3\)
1. \({P}^{1-\gamma}{T}^{\gamma}= \text{constant}\)
2. \({PV}^{\gamma}=\text{constant}\)
3. \({TV}^{\gamma-1}= \text{constant}\)
4. \({P}^{\gamma} {T}^{1-\gamma}=\text{constant}\)
One mole of an ideal gas goes from an initial state \(A\) to the final state \(B\) with two processes. It first undergoes isothermal expansion from volume \(V\) to \(3V\) and then its volume is reduced from \(3V\) to \(V\) at constant pressure. The correct \((P-V)\) diagram representing the two processes is:
| 1. |  |
2. | 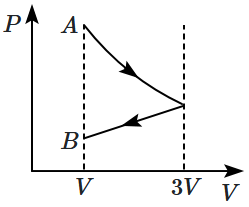 |
| 3. |  |
4. |  |
At a pressure of \(2\) atmospheres, a mass of diatomic gas \((\gamma = 1.4)\), is compressed adiabatically, causing its temperature to rise from \(27^{\circ}\mathrm{C}\) to \(927^{\circ}\mathrm{C}\). The pressure of the gas in the final state is:
1. 8 atm
2. 28 atm
3. 68.7 atm
4. 256 atm
A monoatomic gas at pressure P1 and volume V1 is compressed adiabatically to 1/8th its original volume. What is the final pressure of the gas?
1. P1
2. 16 P1
3. 32 P1
4. 64 P1
In thermodynamic processes which of the following statements is not true?
| 1. | In an adiabatic process, the system is insulated from the surroundings. |
| 2. | In an isochoric process, pressure remains constant. |
| 3. | In an isothermal process, the temperature remains constant. |
| 4. | In an adiabatic process \(PV^\gamma=\mathrm{constant}.\) |




