The correct order of the acidic strength for the above compounds is -
 |
 |
 |
| (I) | (II) | (III) |
1. I > II > III
2. III > I > II
3. II > III > I
4. I > III > II
The most acidic compound among the following is:
| 1. | ClCH2-CH2OH | 2. |  |
| 3. |  |
4. |  |
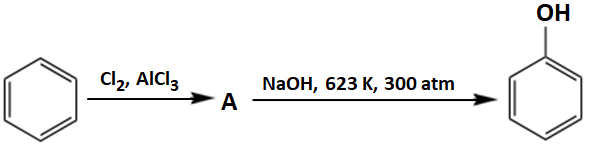
"A" in the reaction below is:
| 1. | Chlorobenzene | 2. | 1,2-Dichlorobenzene |
| 3. | 1,3-dichlorobenzene | 4. | None of these |
Ortho nitrophenol is more acidic than ortho methoxy phenol because:
| 1. | The NO2-group increases the electron density in phenol while the methoxy group decreases the electron density in phenol |
| 2. | The nitro-group is an electron-withdrawing group while the methoxy group is an electron-releasing group |
| 3. | The methoxy-group is an electron-withdrawing group while the nitro group is an electron-releasing group |
| 4. | None of the above |
The product 'B' in the below mentioned reaction is-
1. Phenol
2. Benzol
3. Sodium benzoate
4. Benzal
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Potassium permanganate reagent is used for -
1. Oxidation of primary alcohol to carboxylic acid.
2. Oxidation of primary alcohol to aldehyde.
3. Bromination of phenol to 2,4,6-tribromophenol.
4. Dehydration of propan-2-ol to propene.
The most acidic compound among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | ||
| A. |  |
1. | Phenetole |
| B. |  |
2. | o-Cresol |
| C. |  |
3. | Catechol |
| D. |  |
4. | Resorcinol |
Codes
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |
The ionisation constant of phenol is higher than that of ethanol because -
1. Phenoxide ion is bulkier than ethoxide
2. Phenoxide ion is a stronger base than ethoxide
3. Phenoxide ion is stabilised through delocalisation
4. Phenoxide ion is less stable than ethoxide