The correct statement based on the graph below is:
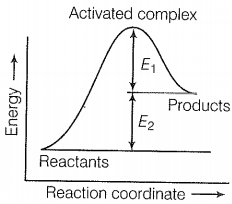
1.
The activation energy of the forward reaction is E1 + E2 and the product is less stable than reactant.
2.
The activation energy of the forward reaction is E1 + E2 and the product is more stable than the reactant.
3.
The activation energy of both forward and backward reaction is E1 + E2 and reactant is more stable than the product.
4.
The activation energy of the backward reaction is E1 and the product is more stable than reactant.

Consider the first-order gas-phase decomposition reaction given below.
A(g) → B(g) + C(g)
The initial pressure of the system before the decomposition of A was . After the lapse of time t, the total pressure of the system increased by X units and became . The rate constant k for the reaction is:
1.
2.
3.
4.
The correct graphical representation of relation between ln k and 1/T is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Consider the Arrhenius equation given below and choose the correct option:
| 1. | Rate constant increases exponentially with increasing activation energy and decreasing temperature. |
| 2. | Rate constant decreases exponentially with increasing activation energy and increasing temperature. |
| 3. | Rate constant increases exponentially with decreasing activation energy and decreasing temperature. |
| 4. | Rate constant increases exponentially with decreasing activation energy and increasing temperature. |
True statement among the following is:
| 1. | The rate of a reaction decreases with the passage of time as the concentration of reactants decreases. |
| 2. | The rate of a reaction is the same at any time during the reaction. |
| 3. | The rate of a reaction is independent of temperature change. |
| 4. | The rate of a reaction decreases with an increase in the concentration of the reactants. |
The correct expression for the rate of reaction given below is:
\(5 \mathrm{Br}^{-}(\mathrm{aq})+\mathrm{BrO}_3^{-}(\mathrm{aq})+6 \mathrm{H}^{+}(\mathrm{aq}) \rightarrow 3 \mathrm{Br}_2(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\mathrm{l})\)
| 1. | \(\frac{\Delta\left[B r^{-}\right]}{\Delta t}=5 \frac{\Delta\left[H^{+}\right]}{\Delta t} \) | 2. | \(\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta t}=\frac{6}{5} \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t} \) |
| 3. | \(\frac{\Delta[\mathrm{Br^-}]}{\Delta t}=\frac{5}{6} \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t} \) | 4. | \(\frac{\Delta\left[\mathrm{Br}^{-}\right]}{\Delta t}=6 \frac{\Delta\left[\mathrm{H}^{+}\right]}{\Delta t}\) |
The correct representation of an exothermic reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. | Both 1 and 2 |
Rate law for the reaction \(A+2 B \rightarrow C\) is found to be
Rate = k[A][B]
If the concentration of reactant 'B' is doubled, keeping the concentration of A constant, then the value of the rate of the reaction will be:
| 1. | The same. | 2. | Doubled. |
| 3. | Quadrupled. | 4. | Halved. |
An incorrect statement about the collision theory of chemical reaction is:
| 1. | It considers reacting molecules or atoms to be hard spheres and ignores their structural features. |
| 2. | The number of effective collisions determines the rate of reaction. |
| 3. | The collision of atoms or molecules possessing sufficient threshold energy results in product formation. |
| 4. | Molecules should collide in the proper orientation for the collision to be effective with sufficient threshold energy and proper orientation. |
A first-order reaction is 50 % completed in 1.26 x 1014 s.
The time required for 100 % completion
of the reaction will be:
1. 1.26 × 1015 s
2. 2.52 × 1014 s
3. 2.52 × 1028 s
4. Infinite


