Match the laws given in Column I with expressions given in Column II.
| Column I | Column II |
| A. Raoult’s law | 1. = CRT |
| B. Osmotic pressure | 2. |
| C. Elevation of boiling point | 3. |
| D. Depression in freezing point | 4. |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 3 | 1 | 4 | 2 |
The molality of a solution containing a certain solute, if there is a freezing point depression of 0.184 °C, is-
(Kf = 18.4)
1. 0.01 m
2. 10 m
3. 0.05 m
4. 100 m
The freezing point of depression constant (Kf ) of benzene is 5.12 K kg mol-1. The freezing point depression for the solution of molality 0.078 m containing a non-electrolyte solute in benzene is-
(rounded off upto two decimal places):
1. 0.80 K
2. 0.40 K
3. 0.60 K
4. 0.20 K
When 20 g of naphthoic acid (C11H8O2) is dissolved in 50 g of benzene (Kf = 1.72 K kg mol–1),
a freezing point depression of 2 K is observed. The van’t Hoff factor (i) is –
| 1. | 0.5 | 2. | 1 |
| 3. | 2 | 4. | 3 |
Freezing point of an aqueous solution is -0.166C. Elevation of boiling point of same solution would be-
(Kb = 0.512 K m-1 and Kf = 1.66 K m-1)
| 1. | 0.18°C | 2. | 0.05°C |
| 3. | 0.09°C | 4. | 0.23°C |
In comparison to a 0.01 M solution of glucose, the depression in the freezing point of a 0.01 M MgCl2 solution will be -
| 1. | Same | 2. | About twice |
| 3. | About three times | 4. | About six times |
A solution containing 6.8 g of a non-ionic solute in 100 g of water was found to freeze at −0.93 oC. The freezing point depression constant of water is 1.86. The molecular weight of the solute is-
| 1. | 13.6 m | 2. | 34 m |
| 3. | 68 m | 4. | 136 m |
The freezing point depression constant for water is 1.86 oC m-1. If 5.00 g Na2SO4 is dissolved in 45.0 g H2O, the freezing point is changed by -3.82 oC. The van’t Hoff factor for Na2SO4 is:
| 1. | 2.63 | 2. | 3.11 |
| 3. | 0.381 | 4. | 2.05 |
The graph, amongst the following, that represents depression in the freezing point is:
| 1. |  |
| 2. | 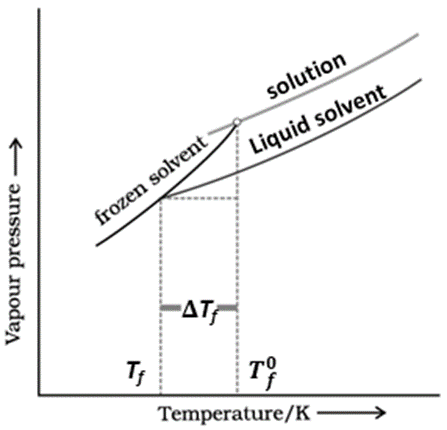 |
| 3. |  |
| 4. | None of the above |
A solution of sucrose (molar mass = 342 g mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water.
The freezing point of the solution obtained will be:
(Kf for water = 1.86 K kg mol-1)
1. -0.372 C
2. 0.372 C
3. 0.572 C
4. -0.572 C


