Which among the following state functions is an extensive property of the system?
1.
Temperature
2.
Volume
3.
Refractive index
4.
Viscosity
Which, among the following, is not a state function?
1. Internal energy
2. Free energy
3. Work
4. Enthalpy
Which of the following is correct for isothermal expansion of an ideal gas -
1. Wrev = Wirr
2. Wrev + Wirr = 0
3. Wrev > Wirr
4. qrev = qirr
The correct statement for a reversible process in a state of equilibrium is:
1. G = – 2.30RT log K
2. G = 2.30RT log K
3. Go = – 2.30RT log K
4. Go = 2.30RT log K
From the following bond energies:
H—H bond energy: 431.37 kJ mol-1
C=C bond energy: 606.10 kJ mol-1
C—C bond energy: 336.49 kJ mol-1
C—H bond energy: 410.50 kJ mol-1
Enthalpy for the reaction,
will be:
| 1. | 1523.6 kJ mol-1 | 2. | -243.6 kJ mol-1 |
| 3. | -120.0 kJ mol-1 | 4. | 553.0 kJ mol-1 |
The values of ΔH and ΔS for the given reaction are 170 kJ and 170 JK-1, respectively.
C(graphite) + CO2(g)→2CO(g)
This reaction will be spontaneous at:
1. 710 K
2. 910 K
3. 1110 K
4. 510 K
1. 1.968 V
2. 2.0968 V
3. 1.0968 V
4. 0.0968 V
The bond energies of , C-H, H-H, and C=C are 198, 98, 103 and145 kcal respectively.
The enthalpy change of the reaction would be-
1. 48 kcal
2. 96 kcal
3. -40 kcal
4. -152 kcal
For the reaction, , at 298K is 164 kJ mol-1. The of the reaction is-
1. \(166.5 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
2. \(141.5 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
3. \(104.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
4. \(-169 \mathrm{~kJ} \mathrm{~mol}^{-1}\)
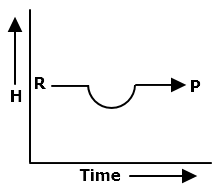
Which plot represents an exothermic reaction?
1.
2.
3.
4.