| Assertion (A): | For an isothermal physical process, the change in internal energy is equal to zero. |
| Reason (R): | The internal energy is directly proportional to temperature for physical processes. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Enthalpy & Internal energy |
82%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Assertion(A): \(\Delta H\) and \(\Delta E\) are almost the same for the reaction, \(N_{2(g)} + O_{2(g)} \rightleftharpoons 2NO_(g)\)
Reason(R): All reactants and products are gases.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Reason(R): All reactants and products are gases.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Subtopic: Enthalpy & Internal energy |
62%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
| Assertion (A): | The increase in internal energy \((\Delta E ) \) for the vapourisation of one mole of water at 1 atm and 373 K is zero. |
| Reason (R): | For all isothermal processes, \(\Delta E = 0 \). |
| 1. | Both A and R are true and R is the correct explanation of A. |
| 2. | Both A and R are true but R is not the correct explanation of A. |
| 3. | A is true and R is false. |
| 4. | A and R both are false. |
Subtopic: Enthalpy & Internal energy |
76%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
| Assertion (A): | Absolute values of the internal energy of substances cannot be determined. |
| Reason (R): | It is impossible to determine the exact values of constituent energies of the substances. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Enthalpy & Internal energy |
80%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Assertion(A): Heat energy is completely transformed into work during the isothermal expansion of a gas.
Reason(R): During an isothermal process, the changes in the internal energy of a gas due to a decrease in pressure are nullified by the changes due to an increase in volume.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Reason(R): During an isothermal process, the changes in the internal energy of a gas due to a decrease in pressure are nullified by the changes due to an increase in volume.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Subtopic: First Law of Thermodynamics | Enthalpy & Internal energy |
55%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Select the correct option based on statements below:
| Assertion (A): | ∆H is positive for endothermic reactions. |
| Reason (R): | If the total enthalpy of reactants and products are Hr & Hp respectively, then for an endothermic reaction HR < HP. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Enthalpy & Internal energy |
77%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Select the correct option based on statements below:
| Assertion (A): | Cdiamond → Cgraphite ∆H and ∆U are the same for this reaction. |
| Reason (R): | Entropy increases during the conversion of diamond to graphite. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Enthalpy & Internal energy |
63%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
For one mole of an ideal gas, which of these statements must be true?
(I) U and H each depend only on temperature.
(II) Compressibility factor z is not equal to 1.
(III) CP, m – CV, m = R
(IV) dU = CVdT for any process.
1. (I), (III) and (IV)
2. (II), (III) and (IV)
3. (III) and (IV)
4. (I) and (III)
Subtopic: Enthalpy & Internal energy |
57%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
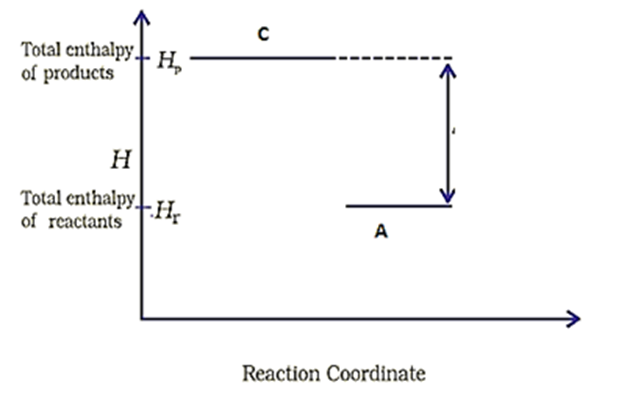
Consider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above
Subtopic: Enthalpy & Internal energy |
81%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Consider the following diagram for a reaction
The nature of the reaction is-
| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of the above |
Subtopic: Enthalpy & Internal energy |
81%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh