As the temperature increases, average kinetic energy of molecules increases. With increase in temperature at constant volume, the pressure -
1. Increases
2. Decreases
3. Remains same
4. Becomes half
Gases possess characteristic critical temperature which depends upon the magnitude of intermolecular forces between the particles. Following are the critical temperatures of some gases.
| Gases | H2 | He | O2 | N2 |
| The critical temperature in Kelvin | 33.2 | 5.3 | 154.3 | 126 |
From the above data, the order of liquefaction of the given gases is-
(write the order from the gas liquefying first)
1.
2.
3.
4.
The SI unit of viscosity coefficient is-
1. Pascal
2. Nsm-2
3. km-2s
4. Nm-2
Atmospheric pressure recorded in different cities are as follows
| Cities | p in N/m2 |
| Shimla | |
| Bangalore | |
| Delhi | |
| Mumbai |
The liquid will boil first at -
1. Shimla
2. Bangalore
3. Delhi
4. Mumbai
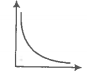
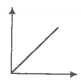
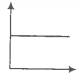
In the given figure, the curve representing an ideal gas is-
1. B only
2. C and D only
3. E and F only
4. A and B only
Increase in kinetic energy can overcome intermolecular forces of attraction. How will increase in temperature effect the viscosity of liquid-
1. Increase
2. No effect
3. Decrease
4. No regular pattern will be followed
With increase in temperature the surface tension of a liquid will:
1. Remains same
2. Decreases
3. Increases
4. No regular pattern is followed
Match the graphs between the following variables with their names.
|
Graphs |
Names |
|
A. Pressure vs temperature graph at constant molar volume |
1. Isotherms |
|
B. Pressure vs |
2. Constant temperature Curve |
|
C. Volume vs temperature graph at constant pressure. |
3. Isochores |
|
4. Isobars |
Codes
| Options: | A | B | C |
| 1. | 3 | 1 | 4 |
| 2. | 1 | 2 | 3 |
| 3. | 5 | 4 | 3 |
| 4. | 4 | 5 | 3 |
Match the following gas law with the equation representing them.
|
A. Boyle’s law |
1. at constant T and p |
|
B. Charle’s law |
2. …constant T,V |
|
C. Dalton’s law |
3. constant |
|
D. Avogadro’s law |
4. at constant n and p |
|
5. at constant n and T |
Codes
| Options: | A | B | C | D |
| 1. | 5 | 4 | 2 | 1 |
| 2. | 3 | 4 | 2 | 1 |
| 3. | 5 | 4 | 3 | 2 |
| 4. | 4 | 5 | 3 | 2 |
Match the following graphs of an ideal gas with their coordinates.
|
Graphical representation |
X and Y coordinates |
|
A. |
1. pV vs V |
|
B. |
2. p vs V |
|
C. |
3. p vs |
Codes
| Options: | A | B | C |
| 1. | 2 | 3 | 1 |
| 2. | 1 | 2 | 3 |
| 3. | 3 | 2 | 1 |
| 4. | 1 | 3 | 1 |