An open flask containing air is heated from 300 K to 500 K. The percentage of air that will escape into the atmosphere if pressure is kept constant is-
1. 80
2. 40
3. 60
4. 20
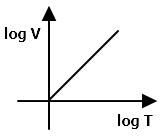
V versus T curves at constant pressure P1 and P2 for an ideal gas are as shown in the figure. The correct statement is-
1. P1>P2
2. P1<P2
3. P1=P2
4. All of the above
The kinetic energy for 14 grams of nitrogen gas at 127°C is nearly -
(mol. mass of nitrogen = 28 and gas constant = 8.31JK–1mol–1)
1. 1.0 J
2. 4.15 J
3. 2493 J
4. 3.3 J
A mixture of CO and CO2 is found to have a density 1.5 g/litre at 30oC and 730 mm. The composition of the mixture is-
1. CO: 44%; CO2 = 56%
2. CO: 32%; CO2 = 68%
3. CO: 70%; CO2 = 30%
4. CO: 9%; CO2 = 91%
A collapsed polythene bag of 30 litre capacity is partially blown up by the addition of 10 litre of N2 at 0.965 atm at 298 K. Subsequently, enough O2 is pumped into bag so that at 298 K and external pressure of 0.990 atm, the bag contains full 30 litre. The final pressure of O2 in the following experiment is-
1. 0.67 atm
2. 0.52 atm
3. 85 atm
4. 20 atm
If density of vapours of a substance of molar mass 18 g/mole at 1 atm pressure and 500 K is
0.36 kg m–3 , then the value of Z for the vapours is-
(R = 0.082 L atm mole K–1)
1.
2.
3. 1.1
4. 0.9
Flask A of volume 10 litre containing 20 gram of H2 and flask B of volume 10 litre containing 88 gram CO2 are connected by a connector having negligible volume. When the valve of the connector is opened, the composition of H2 gas in flask B after opening the valve is-
1. 10%
2. 13%
3. 15%
4. 20%
At a certain temperature for which RT = 25 lit. atm. mol–1, the density of a gas, in gm lit–1, is d = 2.00P + 0.020 P2, where P is the pressure in atmosphere. The molecular weight of the gas in gm mol–1 is-
1. 25
2. 50
3. 75
4. 100
A mixture of carbon monoxide and carbon dioxide is found to have a density of 1.7 g/L at S.T.P. The mole fraction of carbon monoxide is-
1. 0.37
2. 0.40
3. 0.30
4. 0.50