In the nitrogen family, the H-M-H bond angle in the hybrides MH3 gradually becomes closer to 90o if we move from N to Sb. This shows that gradually
1.
The basic strength of hybrides increases.
2.
Pure p-orbitals (almost) are used for M-H bonding.
3.
The bond energies of the M-H bond increase.
4.
The bond pairs of electrons become nearer to the central atom.
Assertion : In trimethyalamine (N(CH3)3), the nitrogen has a pyramidal geometry, wheres in trisilylamine (N(SiH3)3), it has a planer geometry.
Reason : In trimethylamine, nitrogen shows sp3 hybridization while in trisilyamine it shows sp2 hybridization.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
Assertion : Most of the known noble gas compounds are those of xenon.
Reason : The ionisation enthalpy of xenon is very close to that of oxygen.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
Assertion : ClF3 exists but FCl3 does not.
Reason : FCl3 is not formed due to the inability of F to expand its coordination number due to the absence of a vacant d subshell.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
Assertion: Among hydrides of the elements of group 16, water shows unusual physical properties.
Reason: Water shows unusual physical properties due to hydrogen bonding between the water molecules.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
Assertion : Fluorine is a stronger oxidizing agent than chlorine.
Reason : Fluorine can displace chlorine from chlorides.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
The shape and the type of hybridization (of the Cl-atom) in the CIF3 molecule
| 1. | 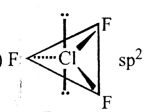 |
2. |  |
| 3. |  |
4. |  |
A yellow metallic powder is burnt in a stream of fluorine to obtain a colourless gas X which is thermally stable and chemically inert. Its molecule has octahedral geometry. Another colourless gas Y with same constituent atoms as that of X is obtained when sulphur dichloride is heated with sodium fluoride. Its molecule has triangular pyramidal structure X and Y are
(1) SF4 and S2F2
(2) SF6 and SF4
(3) NaF and NaCl
(4) SF4 and SF6
Oxide of a non-metal possesses the following characterstics.
(i) It is both a proton donor and proton acceptor
(ii) It is poor conductor of electricity
(iii) It reacts readily with basic and acidic oxides
(iv) It oxidises Fe at boiling point.
The oxide is
(1) H2O
(2) CO2
(3) H2O2
(4) NO
Strong oxidising agent and strong reducing agents are respectively
(1) F2, HI
(2) Cl2, HCl
(3) Br2,HBr
(4) I2, HF






