A closed vessel contains equal number of nitrogen and oxygen molecules at pressure of P mm. If nitrogen is removed from the system, then the pressure will be
(1)
(2)
(3)
(4)
Two vessels of capacities 3 litres and 4 litres are separately filled with a gas. The pressures are respectively 202 kPa and 101 kPa. The two vessels are connected. The gas pressure will be now, at constant temperature.
(1) 151.5 kPa
(2) 144 kPa
(3) 303 kPa
(4) 175 kPa
2 gms of hydrogen diffuses from a container in 10 minutes. How many gms of oxygen would diffuse through the same time under similar conditions?
1. 0.5 gm
2. 4 gm
3. 6 gm
4. 8 gm
The temperature of a sample of gas is raised from 127ºC to 527ºC. The average kinetic energy of the gas
(1) Does not changes
(2) Is doubled
(3) Is halved
(4) Cannot be calculated
A helium atom is two times heavier than a hydrogen molecule at 298 K, the average kinetic energy of helium is
(1) Two times that of hydrogen molecules
(2) Same as that of hydrogen molecules
(3) Four times that of hydrogen molecules
(4) Half that of hydrogen molecules
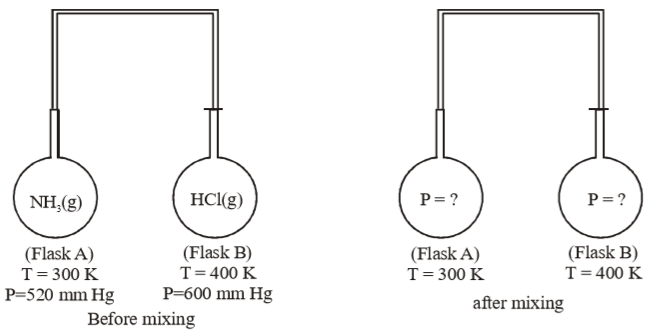
Two flasks A and B of equal volume containing and HCl gases, are connected by a narrow tube of negligible volume. The two gases were prevented from mixing by stopper fitted in connecting tube. For further detail of experiment refer to the given figure. What will be final pressure in each flask when passage connecting two tubes are opened. Assume ideal gas behaviour of and gas and the reaction.
(1) 40 mm Hg
(2) 60 mm Hg
(3) 20 mm Hg
(4) 10 mm Hg
The ratio of average molecular kinetic energy of to that of , both at 300 K is
(1) 1 : 1
(2) 7 : 2
(3) 176 : 1
(4) 2 : 7
A mono atomic gas diatomic gas and triatomic gas are mixed, taking one mole of each for the mixture is
(1) 1.40
(2) 1.428
(3) 1.67
(4) 1.33
According to kinetic theory of gases, for a diatomic molecule
(1) The pressure exerted by the gas is proportional to the mean velocity of the molecule.
(2) The pressure exerted by the gas is proportional to the root mean square velocity of the molecule.
(3) The root mean square velocity of the molecule is inversely proportional to the temperature.
(4) The mean translational kinetic energy of the molecule is proportional to the absolute temperature.
The values of vander waals constant ‘a’ for the gases , , and are 1.36, 1.39, 4.17 and 2.253 atom mol-2 respectively. The gas which can most easily be liquefied is
(A)
(B)
(C)
(D)