For a certain reaction of order \('n'\) the time for half change \(t_{1/2}\) is given by; 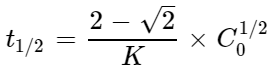 \(=,t_{1/2}=\dfrac{2-\sqrt2}{K}\times C^{1/2}_0,\) where \(K\) is rate constant and \(C_0\) is initial concentration. The value of \(n\) is:
\(=,t_{1/2}=\dfrac{2-\sqrt2}{K}\times C^{1/2}_0,\) where \(K\) is rate constant and \(C_0\) is initial concentration. The value of \(n\) is:
1. 1
2. 2
3. 0
4. 0.5
In the following first order competing reactions:
A + Reagent Product
B + Reagent Product
The ratio of K1/K2 if only 50% of B will have been reacted when 94% of A has been reacted in same time is:
1. 4.06
2. 0.246
3. 2.06
4. 0.06
For a reversible reaction A B, Ist order in both the directions, the rate of reaction is given by:
1. K1[A]
2. -K2B
3. K1[A] + K2[B]
4. K1[A] - K2[B]
The time for half-life of a first order reaction is 1 hr. What is the time taken for 87.5% completion of the reaction?
1. 1 hour
2. 2 hour
3. 3 hour
4. 4 hour
Which order of reaction obeys the relation t1/2 = 1/Ka?
1. First
2. Second
3. Third
4. Zero
The chemical reaction, 2O3 3O2 proceeds as follows;
O3 O2 + O .....(Fast)
O+O3 2O2 ....(Slow)
The rate law expression should be:
1. r = K[O3]2
2. r = K[O3]2[O2]-1
3. r = K[O3][O2]
4. unpredictable
The rate of reaction becomes 2 times for every 10°C rise in temperature. How the rate of reaction will increase when temperature is increased from 30°C to 80°C?
1. 16
2. 32
3. 64
4. 128
The rate constant (K) for the reaction 2A +B Product was found to be 2.5x10-5 litre mol-1 sec-1 after 15 sec, 2.60 x10-5 litre mol-1sec-1 after 30 sec and 2.55 x10-5litre mol-1 sec-1 after 50 sec. The order of reaction is:
1. 2
2. 3
3. zero
4. 1
A reaction A2 + B2 2AB occurs by the following mechanism:
A2 A + A .....(slow)
A + B2 AB + B .....(fast)
A + B AB .....(fast)
Its order would be:
1. 3/2
2. 1
3. 0
4. 2
For a given reaction, the presence of a catalyst reduces the energy of activation by 2 kcal at 27 oC. The rate of reaction will be increased by:
1. 20 times
2. 14 times
3. 28 times
4. 2 times






