For a non-stoichiometric reaction 2A + B C +D, the following kinetic data were obtained in three separate experiments, all at 298 K.
Initial
Concentration (A)
Initial
Concentration (B)
Initial rate of formation of C (mol L-1 S-1)
0.1 M
0.1 M
0.1 M
0.2 M
0.2 M
0.1 M
The rate law for the formation of C is:
1. d[C]/dt = k[ A][B]
2. d[C]/dt = k[ A]2[B]
3. d[C]/dt = k[ A][B]2
4. d[C]/dt = k[ A]
Initial
Concentration (A)
Concentration (B)
For the reaction N2 + 3H2 2NH3, the rate = 2 x 10-4 M s-1 .Therefore, the rate is given as:
1. 10-4 Ms-1
2. 104 Ms-1
3. 10-2 sM-1
4. 10-4 sM-1
If 'I' is the intensity of absorbed light and 'c' is the concentration of AB for the photochemical process AB + hv→ AB *, the rate of formation of AB * is directly proportional to:
1. c
2. I
3. I2
4. cI
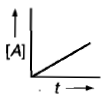
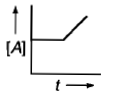
Which curve represents zero order reaction?
1. 

2. 
3. 
4. 
In a reaction, the rate expression is, rate = K[A][B]2/3[C]0, the order of the reaction is:
1. 1
2. 2
3. 5/3
4. zero
The rate of a reaction get doubles when the temperature changes from 7°C to 17°C. By what factor will it change for the temperature change from 17°C to 27°C?
1. 1.81
2. 1.71
3. 1.91
4. 1.76
In the Arrhenius equation K = Ae-Ea/RT, the quantity e-Ea/kT is referred as:
1. Boltzmann factor.
2. Frequency factor.
3. Activation factor.
4. None of the above.
For the elementary step, (CH3)3.CBr(aq) → (CH3)3C+ (aq) + Br- (aq), the molecularity is:
1. Zero
2. 1
3. 2
4. Cannot be ascertained
When ethyl acetate was hydrolysed in pressure of 0.1 N HCl, the rate constant was found to be 5.40 x 10-5 sec-1 . But when 0.1 N H2SO4 was used for hydrolysis, the rate constant was found to be 6.25 X10-5sec-1. Thus, it may be concluded that:
1. H2SO4 is stronger than HCI
2. H2SO4 is weaker than HCl
3. H2SO4 and HCl both have the same strength
4. The data are not sufficient to compare the strength of H2SO4 and HCI
The half time of a second order reaction is:
1. Inversely proportional to the square of the initial concentration of the reactants.
2. Inversely proportional to the initial concentration of the reactants.
3. Proportional to the initial concentration of reactants.
4. Independent of the initial concentration of reactants.