Following mechanism has been proposed for a reaction,
2A+B D+E
A+B C+D ...(Slow)
A+ C E ...(Fast)
The rate law expression for the reaction is:
1. r = K[A]2[B]
2. r=K[A][B]
3. r= K[A]2
4. r= K[A][C]
In gaseous reactions important for the understanding of the upper atmosphere H2O and O react bimolecularly to form two OH radicals. H for this reaction is 72kJ at 500 K and Ea is 77 kJ mol-1, then Ea for the bimolecular recombination of two OH radicals to form H2O and O is:
1. 3 kJ mol-1
2. 4 kJ mol-1
3. 5 kJ mol-1
4. 7 kJ mol-1
For a reaction A Product, rate law is . The concentration of A left after time t when is:
1.
2.
3.
4.
For an exothermic chemical process occurring in two steps as;
(i) A+BX(Slow)
(ii) XAB (Fast)
The progress of the reaction can be best described by:
| 1. |  |
2. |  |
| 3. |  |
4. | All of the above. |
For a non-stoichiometric reaction 2A + B C +D, the following kinetic data were obtained in three separate experiments, all at 298 K.
|
Initial Concentration (A) |
Initial Concentration (B) |
Initial rate of formation of C (mol L-1 S-1) |
| 0.1 M | 0.1 M | |
| 0.1 M | 0.2 M | |
| 0.2 M | 0.1 M |
The rate law for the formation of C is:
1. d[C]/dt = k[ A][B]
2. d[C]/dt = k[ A]2[B]
3. d[C]/dt = k[ A][B]2
4. d[C]/dt = k[ A]
For the reaction N2 + 3H2 2NH3, the rate = 2 x 10-4 M s-1 .Therefore, the rate is given as:
1. 10-4 Ms-1
2. 104 Ms-1
3. 10-2 sM-1
4. 10-4 sM-1
If 'I' is the intensity of absorbed light and 'c' is the concentration of AB for the photochemical process AB + hv→ AB *, the rate of formation of AB * is directly proportional to:
1. c
2. I
3. I2
4. cI
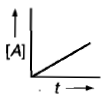
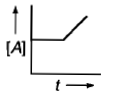
Which curve represents zero order reaction?
1. 

2. 
3. 
4. 
In a reaction, the rate expression is, rate = K[A][B]2/3[C]0, the order of the reaction is:
1. 1
2. 2
3. 5/3
4. zero
The rate of a reaction get doubles when the temperature changes from 7°C to 17°C. By what factor will it change for the temperature change from 17°C to 27°C?
1. 1.81
2. 1.71
3. 1.91
4. 1.76