What mass of isobutylene is obtained from 37 g of tertiary butyl alcohol by heating with 20% H2SO4 at 363 K, if the yield is 65%?
1. 16 g
2. 18.2 g
3. 20 g
4. 22 g
Which of the following reaction is used for preparation of glycol industrially?
| 1. | 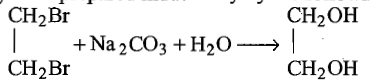  |
| 2. | 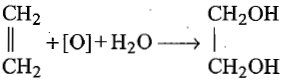  |
| 3. | 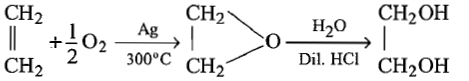  |
| 4. | none of the above |
How many structural isomers of primary alcohols can be formed with the given molecular formula \(\text{C}_5\text{H}_{11}\text{OH}\)?
| 1. | 5 | 2. | 4 |
| 3. | 2 | 4. | 3 |
The solubility of phenol in water is lower. This can be attributed to:
1. Non-polar nature of phenol
2. Acidic nature of -OH group
3. Non-polar hydrocarbon part in phenol
4. None of the above
The decreasing order of boiling points of \(1^\circ\), \(2^\circ\), and \(3^\circ\) alcohol is-
1. \(1^\circ\)> \(2^\circ\)> \(3^\circ\)
2. \(3^\circ\)> \(2^\circ\)> \(1^\circ\)
3. \(2^\circ\) > \(1^\circ\) > \(3^\circ\)
4. None of the above
The boiling points of thio-ethers are .... than those of ether.
1. lesser
2. equal
3. higher
4. none of these
The most effective reagent used to converts but-2-enal to but-2-enol is-
1. KMnO4
2. NaBH4
3. H2/Pt
4. K2Cr2O7/H2SO4
23g of sodium react with CH3OH to give-
1. 1 mole of O2
2. 1/2 mole of H2
3. 1 mole of H2
4. None of the above
Phenol and benzoic acid can be distinguished by:
1. Aqueous NaHCO3
2. Aqueous NaNO3
3. Aqueous NaOH
4. Conc. H2SO4
Glycerol on oxidation with conc HNO3 gives:
1. tartronic acid
2. mesoxalic acid
3. oxalic acid
4. glyceric acid






