Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly
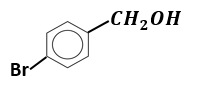
Which of the following compounds is most prone to oxidation?
1.
\(\mathrm{CH}_3-\mathrm{CHOH}-\mathrm{CH}_3\)2.

3.
\(\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{O}-\mathrm{CH}_2-\mathrm{CH}_3\)4.
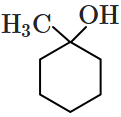
\(\mathrm{CH}_3-\mathrm{CHOH}-\mathrm{CH}_3\)

\(\mathrm{CH}_3-\mathrm{CH}_2-\mathrm{O}-\mathrm{CH}_2-\mathrm{CH}_3\)

Subtopic: Alcohols: Preparation & Properties |
Level 3: 35%-60%
Hints
Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly
Identify the product (P) in the given reaction:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Alcohols: Preparation & Properties |
Level 4: Below 35%
Hints
Which of the following react with HBr at faster rate?
Subtopic: Alcohols: Preparation & Properties |
76%
Level 2: 60%+
Hints
The reagent used in the conversion of the following reaction is:
| 1. | \(\mathrm{NaBH}_4 \) | 2. | \(\mathrm{LiAlH}_4 \) |
| 3. | \(PCC \) | 4. | \(\mathrm{KMnO}_4\) |
Subtopic: Alcohols: Preparation & Properties |
81%
Level 1: 80%+
Hints
The product (A) in the below mentioned reaction is:


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Alcohols: Preparation & Properties |
64%
Level 2: 60%+
Hints
Links
Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly
Identify the major product,
1. 
2. 
3. 
4.
Subtopic: Alcohols: Preparation & Properties |
56%
Level 3: 35%-60%
Hints
Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly

(a)
(b)
(c)
(d)
Subtopic: Alcohols: Preparation & Properties |
59%
Level 3: 35%-60%
Hints
Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly

(a) No reaction
(b)
(c)
(d)
Subtopic: Mechanism of Dehydration, Methanol & Ethanol |
52%
Level 3: 35%-60%
Hints
Consider the following alcohols,
(I) 
(III)
The order of decreasing reactivities of these alcohols towards nucleophilic substitution with HBr is:-
(a) III>I>IV>II
(b) III>I>II>IV
(c) I>III>IV>II
(d) I>III>II>IV
Subtopic: Alcohols: Preparation & Properties | Mechanism of Dehydration, Methanol & Ethanol |
74%
Level 2: 60%+
Hints
The structure of A in the below mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Alcohols: Preparation & Properties |
86%
Level 1: 80%+
Hints