Equal weights of methane and oxygen are mixed in an empty container at 25°C. The fraction of the total pressure exerted by oxygen is
(1) 1/3
(2) 1/2
(3) 2/3
(4)
The rate of diffusion of methane at a given temperature is twice that of gas X. The molecular weight of X is
(1) 64.0
(2) 32.0
(3) 4.0
(4) 8.0
The kinetic energy for 14 grams of nitrogen gas at 127°C is nearly (mol. mass of nitrogen = 28 and gas constant = 8.31JK–1mol–1)
(1) 1.0 J
(2) 4.15 J
(3) 2493 J
(4) 3.3 J
If the average velocity of N2 molecules is 0.3 m/s at 27°C, then the velocity of 0.6 m/s will take place at
(1) 273 K
(2) 927 K
(3) 1000 K
(4) 1200 K
A gas such as carbon monoxide would be most likely to obey the ideal gas law at
(1) high temperatures and low pressures
(2) low temperatures and high pressures
(3) high temperatures and high pressures
(4) low temperatures and low pressures
A gaseous mixture was prepared by taking equal moles of CO and N2. If the total pressure of the mixture was found 1 atmosphere, the partial pressure of the nitrogen (N2) in the mixture is
(a) 0.8 atm
(b) 0.9 aim
(c) 1 atm
(d) 0.5 atm
What volume of oxygen gas (O2) measured at 0°C and 1 atm, is needed to burn completely 1 L of propane gas (C3H8) measured under the same conditions?
(1) 7L
(2) 6L
(3) 5L
(4) 10L
A certain volume of argon gas (Mol. wt. = 40) requires 45 s to effuse through a hole at a certain pressure and temperature. The same volume of another gas of unknown molecular weight requires 60s to pass through the same hole under the same conditions of temperature and pressure. The molecular weight of the gas is
(A) 53
(B) 35
(C) 71
(D) 120
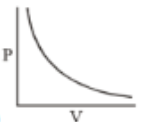
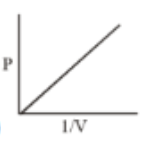
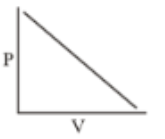
Which of the following curves does not represent Boyle's Law
(1) 
(2)
(3) 
(4) 
The ratio of average molecular kinetic energy of to that of , both at 300 K is
(A) 1 : 1
(B) 7 : 2
(C) 176 : 1
(D) 2 : 7







