

can be repared from williamson's synthesis using
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | All of these |
1. 
2. C6H5CH2Cl and
3. 
4. All of these
Methanol and ethanol can be distinguished by the following:
1. By reaction with metallic sodium
2. By reaction with caustic soda
3. By heating with iodine and washing soda
4. By heating with zinc and inorganic mineral acid
The product 'D' in the below mentioned reaction is:
\(CH_3MgBr + \text{cyclopentanone}\ \)
\(\xrightarrow{H_2O}\ A\ \xrightarrow{HBr}\ B\)
\( B\ \xrightarrow{Mg/Ether}\ C \xrightarrow[H_3O^+]{HCHO}\ D\)
| 1. |  |
2. | 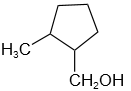 |
| 3. |  |
4. |  |
Consider the following alcohols,
| I. |  |
| II. |  |
| III. |  |
| IV. |  |
Ease of dehydration of the above alcohols will be in the order of:
| 1. | I < II < III < IV | 2. | I > II > III > IV |
| 3. | III < II < I < IV | 4. | II < III < IV < I |
The number of moles of HI
consumed by the given compound is:
| 1. | 1 | 2. | 2 |
| 3. | 3 | 4. | 4 |
R-CH2-CH2OH can be converted into RCH2CH2COOH. The correct sequence of reagents is:
1. PBr3, KCN, H+
2. PBr3, KCN, H2
3. KCN, H+
4. HCN, PBr3, H+
Chlorobenzene reacts with Mg in dry ether to give a compound (A) which further reacts
with ethanol to yield
1. phenol
2. benzene
3. ethyl benzene
4. phenyl ether
Ethyl alcohol is denatured by:
1. Methanol and formic acid
2. KCN
3. CH3OH and C6H6
4. CH3OH and pyridine
The compound B formed in the following sequence of reactions,
CH3CH2CH2OHAB will be:
1. Propyne
2. Propene
3. Propanal
4. Propane
The boiling point of ethanol is higher than that of dimethyl ether due to the presence of
| 1. | H-bonding in ethanol | 2. | H-bonding in dimethyl ether |
| 3. | -CH3 group in ethanol | 4. | -CH3 group in dimethyl ether |








